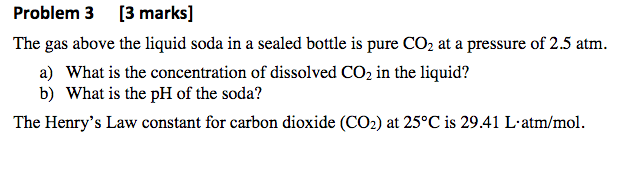1
answer
0
watching
373
views
bluecat630Lv1
6 Feb 2020
A certain soft drink is bottled so that a bottle at 25°C contains CO2 gas at a pressure of 4 atm over the liquid. Assuming the partial pressure of CO2 in the atmosphere is 4.0×10-4 atm, calculate the equilibrium concentration of CO2 in the soda after the bottle is opened. The Henry’s Law constant for CO2 in an aqueous solution is 3.1×10-2 mol/L·atm at 25°C.
A certain soft drink is bottled so that a bottle at 25°C contains CO2 gas at a pressure of 4 atm over the liquid. Assuming the partial pressure of CO2 in the atmosphere is 4.0×10-4 atm, calculate the equilibrium concentration of CO2 in the soda after the bottle is opened. The Henry’s Law constant for CO2 in an aqueous solution is 3.1×10-2 mol/L·atm at 25°C.
Liked by suwilanjisimbeye85 and 1 others
2 Jun 2021