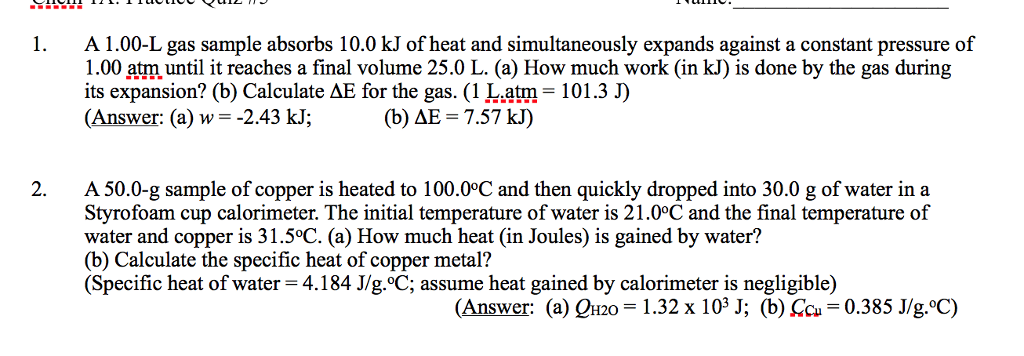1
answer
0
watching
318
views
18 Dec 2019
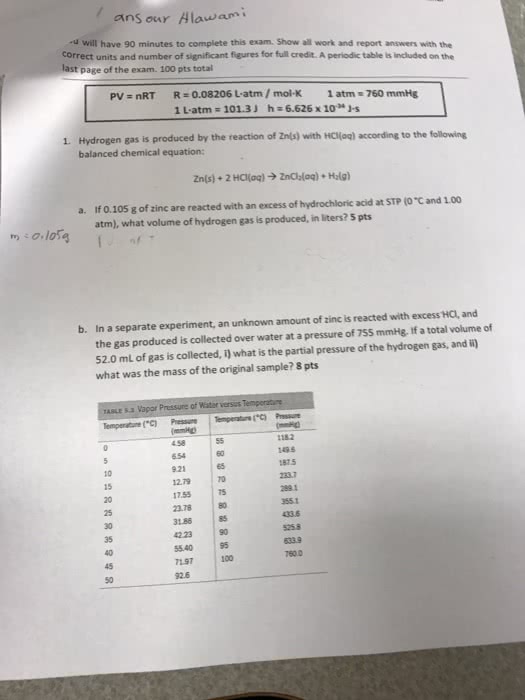
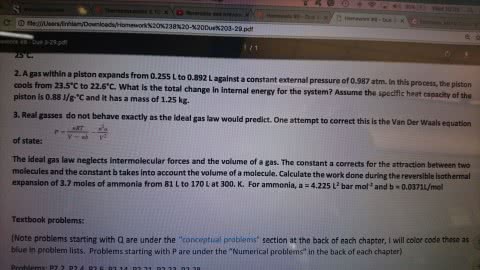
6. One mole of an ideal gas is expanded from a volume of 1.00 liter to a volume of 6.82 liters against a
constant external pressure of 1.00 atm. How much work (in joules) is performed on the
surroundings? Ignore significant figures for this problem. (T = 300 K; 1 L atm = 101.3 J)
A) 691 J
B) 1.77 Ã 10J
C) 295 J
D) 590 J
E) none of these
6. One mole of an ideal gas is expanded from a volume of 1.00 liter to a volume of 6.82 liters against a
constant external pressure of 1.00 atm. How much work (in joules) is performed on the
surroundings? Ignore significant figures for this problem. (T = 300 K; 1 L atm = 101.3 J)
A) 691 J
B) 1.77 Ã 10J
C) 295 J
D) 590 J
E) none of these
1
answer
0
watching
318
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Reid WolffLv2
31 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232