0
answers
0
watching
171
views
18 Dec 2019
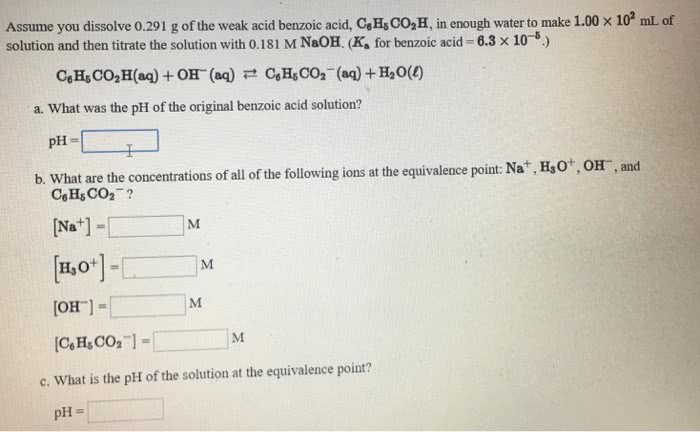
Assume you dissolve 0.235 g of the weak acid benzoic acid, C6H5CO2H, in enough water to make 1.00 â 102 mL of solution and then titrate the solution with 0.138 M NaOH. C6H5CO2H(aq) + OH-(aq) C6H5CO2-(aq) + H2O(â) What are the concentrations of the following ions at the equivalence point? Na+, H3O+, OH- C6H5CO2-
Assume you dissolve 0.235 g of the weak acid benzoic acid, C6H5CO2H, in enough water to make 1.00 â 102 mL of solution and then titrate the solution with 0.138 M NaOH. C6H5CO2H(aq) + OH-(aq) C6H5CO2-(aq) + H2O(â) What are the concentrations of the following ions at the equivalence point? Na+, H3O+, OH- C6H5CO2-