1
answer
0
watching
135
views
18 Dec 2019
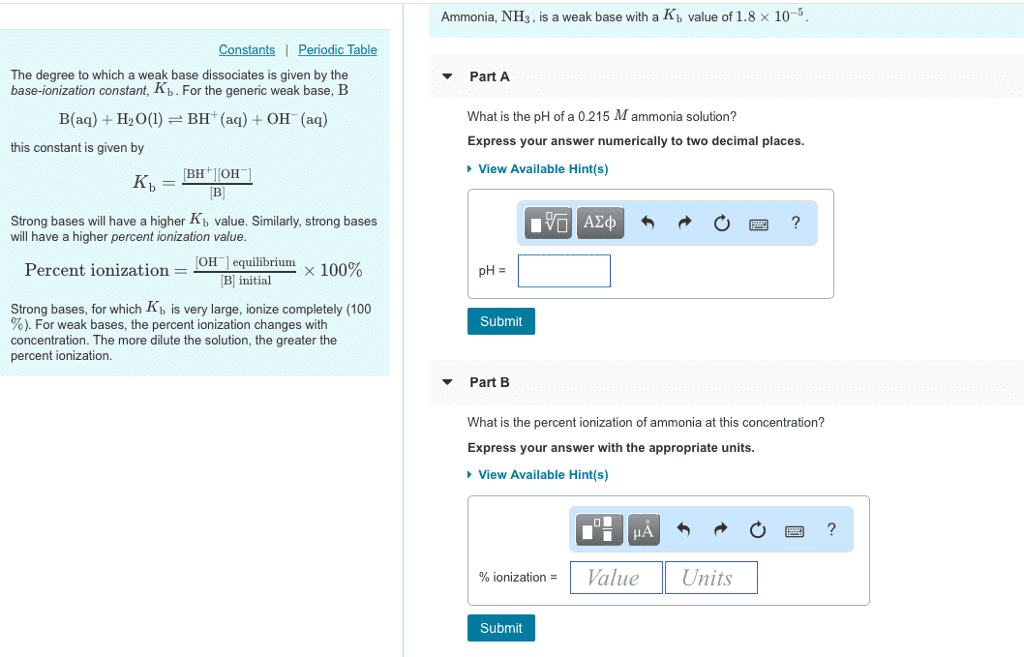
Ammonia (NH3) is a weak base with a Kb of 1.8*10-5. If you have a 0.252 M solution of ammonia, what is the pH.
Hints: write the reaction of ammonia acting as a base with water, write the Kb expression, complete the ICE table, determine the equilibrium conditions. Consider: if ammonia acts as a base and produces OH-, how do you calculate the pH?
Ammonia (NH3) is a weak base with a Kb of 1.8*10-5. If you have a 0.252 M solution of ammonia, what is the pH.
Hints: write the reaction of ammonia acting as a base with water, write the Kb expression, complete the ICE table, determine the equilibrium conditions. Consider: if ammonia acts as a base and produces OH-, how do you calculate the pH?
Sixta KovacekLv2
31 Dec 2019