1
answer
0
watching
186
views
18 Dec 2019
1)What element is being oxidized in the following redox reaction?
Cr(OH)4^-(aq) +ClO^- (aq)----> CrO4^2- (aq) + Cl^-
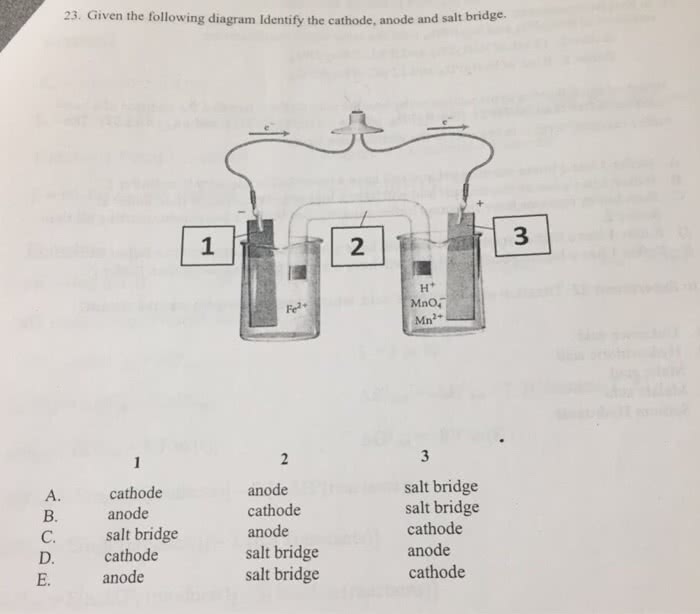
1B) Balance the following redox reaction if it occurs in acidic solution. What are the coefficients in frot of H+ and Fe3+ in the balance reaction?
Fe2+ (aq) + MnO4^- (aq) ---> Fe 3+ (aq) +Mn 2+
1C) determine the cell notation for the redox reaction given below. [hint: remember sometimes inert electrodes are used.
3 Cl2(g) + 2Fe(s) ---> 6Cl- (aq) +2 Fe 3+ (aq)
1)What element is being oxidized in the following redox reaction?
Cr(OH)4^-(aq) +ClO^- (aq)----> CrO4^2- (aq) + Cl^-
1B) Balance the following redox reaction if it occurs in acidic solution. What are the coefficients in frot of H+ and Fe3+ in the balance reaction?
Fe2+ (aq) + MnO4^- (aq) ---> Fe 3+ (aq) +Mn 2+
1C) determine the cell notation for the redox reaction given below. [hint: remember sometimes inert electrodes are used.
3 Cl2(g) + 2Fe(s) ---> 6Cl- (aq) +2 Fe 3+ (aq)
Tod ThielLv2
31 Dec 2019