1
answer
0
watching
138
views
18 Dec 2019
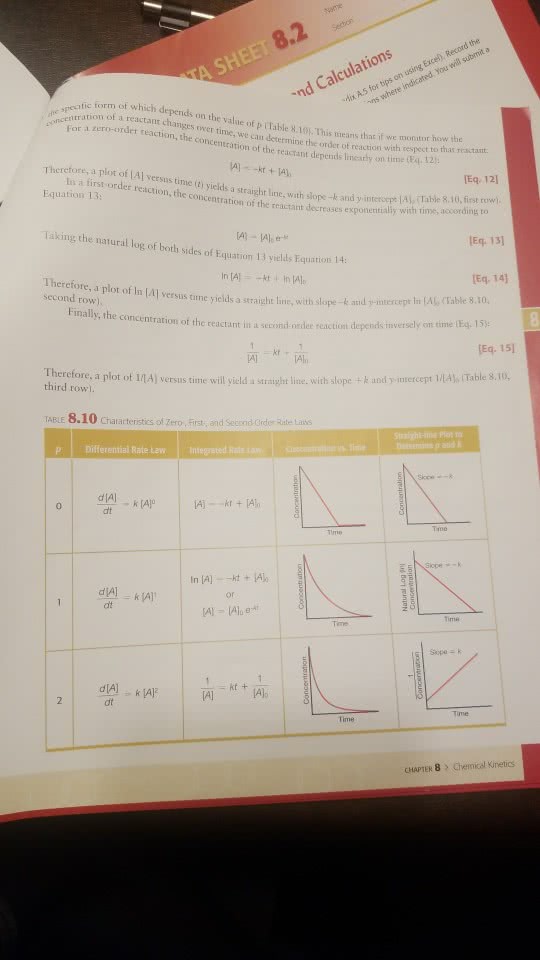
What rate law and integrated rate lets you calculate the time required to let your [CV+] change to half of its value at t=0? Second order rate law and its integrated rate is useful to calculate [CV+] to half life at t =0.
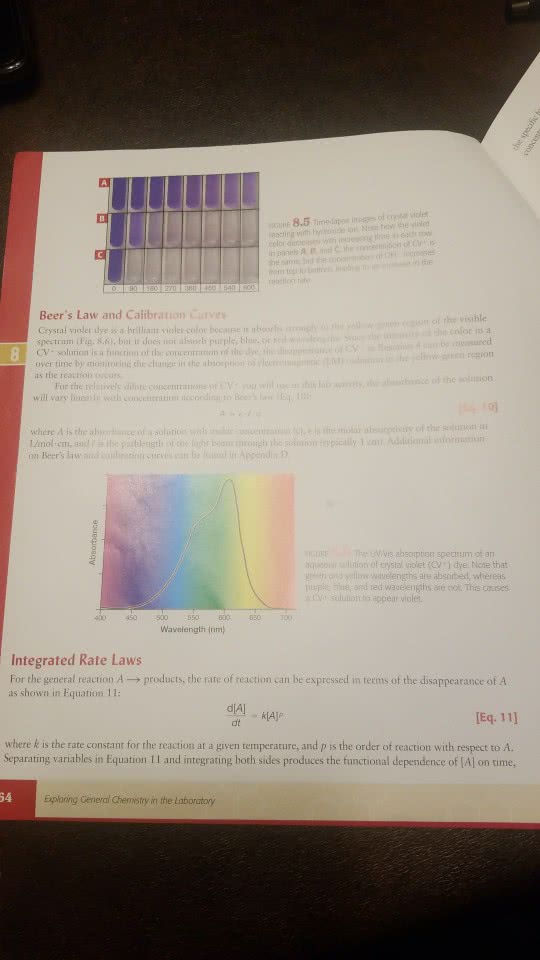
We want to find the rate law that describes how reactant concentrations affect the rate of the reaction shown by: CV+ + OH- --------------- CVOH
The general form of the rate law we will find is: rate = k[CV+]x [OH-]
where x, y are the reaction orders with respect to reactanst, x+y is the overall reaction order and k is the rate constant.
so far, I have found rate = 2.0 * 10-6 and k=1.25.
Now, I need to calculate the time needed for the concentration to be halved.
What rate law and integrated rate lets you calculate the time required to let your [CV+] change to half of its value at t=0? Second order rate law and its integrated rate is useful to calculate [CV+] to half life at t =0.
We want to find the rate law that describes how reactant concentrations affect the rate of the reaction shown by: CV+ + OH- --------------- CVOH
The general form of the rate law we will find is: rate = k[CV+]x [OH-]
where x, y are the reaction orders with respect to reactanst, x+y is the overall reaction order and k is the rate constant.
so far, I have found rate = 2.0 * 10-6 and k=1.25.
Now, I need to calculate the time needed for the concentration to be halved.
Nelly StrackeLv2
31 Dec 2019