1
answer
1
watching
291
views
13 Dec 2019
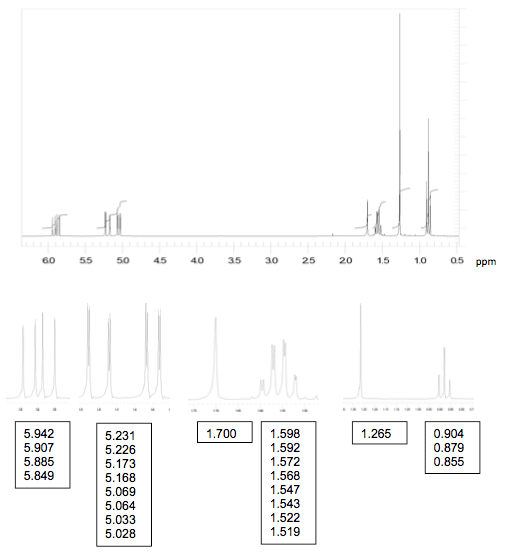
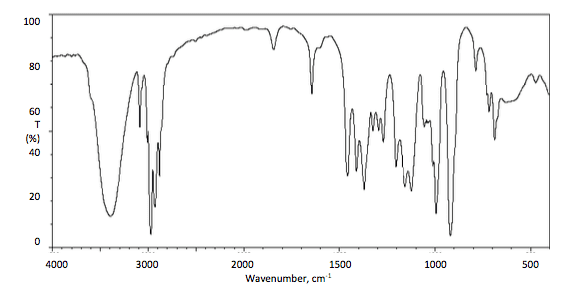
An organic compound A is composed of carbon, hydrogen andnitrogen, with carbon constituting over 60% of the mass. It shows amolecular ion at m/z= 112 in the mass spectrum.
- Write a plausible molecular formula for compound A
- How many rings + double bonds must be present in compoundA?
An organic compound A is composed of carbon, hydrogen andnitrogen, with carbon constituting over 60% of the mass. It shows amolecular ion at m/z= 112 in the mass spectrum.
- Write a plausible molecular formula for compound A
- How many rings + double bonds must be present in compoundA?
Jean KeelingLv2
17 Dec 2019