0
answers
0
watching
142
views
13 Dec 2019
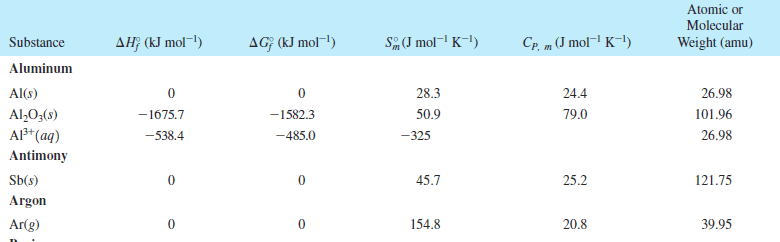
a sample of 1.000 mole of an ideal monatomic gas at 300.0 K and1.00 bar is taken through the following cycle.
a) constant volume heating to twice its initial temperature,followed by
b) reversible adiabatic expansion back to its initial T,followed by
c) reversible isothermal compression back to its initialpressure.
calculate w,q,and delta U (using the ideal gas expression forthe internal energy of a gas) for each of the steps and the overallprocess.
a sample of 1.000 mole of an ideal monatomic gas at 300.0 K and1.00 bar is taken through the following cycle.
a) constant volume heating to twice its initial temperature,followed by
b) reversible adiabatic expansion back to its initial T,followed by
c) reversible isothermal compression back to its initialpressure.
calculate w,q,and delta U (using the ideal gas expression forthe internal energy of a gas) for each of the steps and the overallprocess.