1
answer
0
watching
342
views
13 Dec 2019
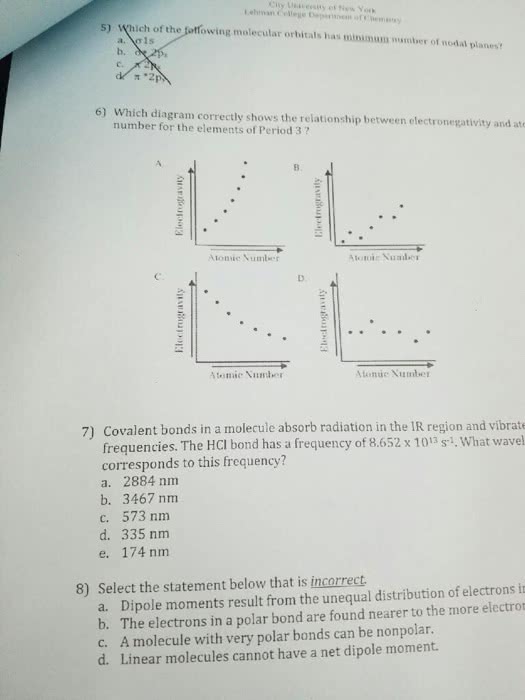
1) Which of the following statments is incorrect?
A)Linear molecules cannot have a net dipole moment
B)Dipole moments result from the unequal distribution of electrons in a molecule
C)the electrons in a polar bond are found nearer to the more electronegative element
D)a molecule with very polar bonds can be nonpolar
E)ionic bonding reults from the transfer of electrons from one atom to another
2)Which metals form cations with vary positive charges?
A)transition metals
B) group 1 metals
C) group 2 metals
D)group 3 metals
E)metalloids
1) Which of the following statments is incorrect?
A)Linear molecules cannot have a net dipole moment
B)Dipole moments result from the unequal distribution of electrons in a molecule
C)the electrons in a polar bond are found nearer to the more electronegative element
D)a molecule with very polar bonds can be nonpolar
E)ionic bonding reults from the transfer of electrons from one atom to another
2)Which metals form cations with vary positive charges?
A)transition metals
B) group 1 metals
C) group 2 metals
D)group 3 metals
E)metalloids
1
answer
0
watching
342
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Irving HeathcoteLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232