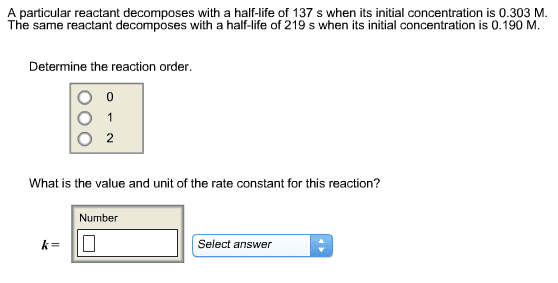
1
answer
0
watching
61
views
13 Dec 2019
1. A first order reaction is seen to go from a reactant concentration of 0.0451 M to 0.0321 M in 41.8 seconds? How long (in seconds) after the reaction starts will the reactant concentration be 0.0180 M? I know the correct answer to this is 113 seconds, but I'm not sure how to get there.
2. HCOOH (g) â CO2 (g) + H2 (g)
At 555.0° C, the half life for this reaction is 23.5 min. How many seconds are needed for the initial concentration of HCOOH to decrease by a factor of 10? The correct answer to this is 4740 seconds, but again not sure how/why.
1. A first order reaction is seen to go from a reactant concentration of 0.0451 M to 0.0321 M in 41.8 seconds? How long (in seconds) after the reaction starts will the reactant concentration be 0.0180 M? I know the correct answer to this is 113 seconds, but I'm not sure how to get there.
2. HCOOH (g) â CO2 (g) + H2 (g)
At 555.0° C, the half life for this reaction is 23.5 min. How many seconds are needed for the initial concentration of HCOOH to decrease by a factor of 10? The correct answer to this is 4740 seconds, but again not sure how/why.
1
answer
0
watching
61
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Casey DurganLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232