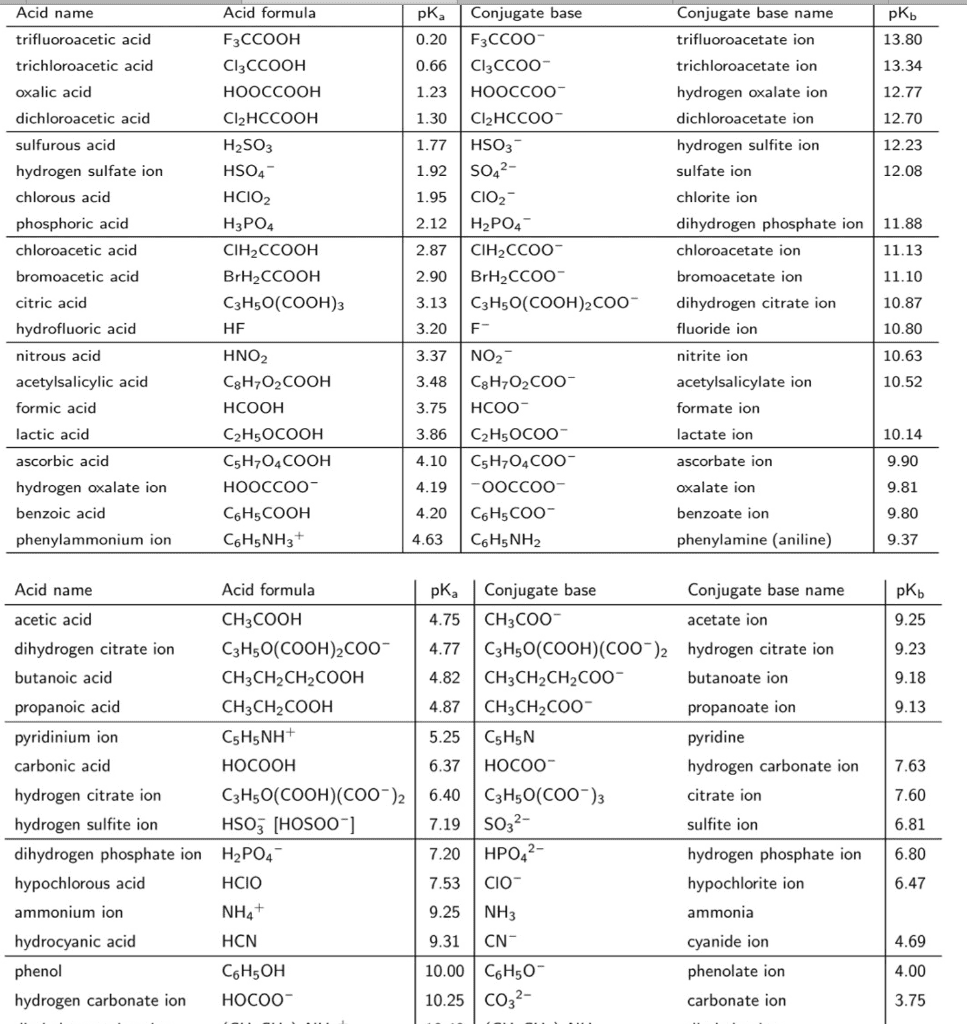
Aqueous solutions of phosphoric acid and sodium nitrite are combined, and the following equilibrium is established. H3PO4(aq) + NO2-(aq) <----> H2PO4-(aq) + HNO2(aq) The equilibrium constant Kc for this reaction is greater than one. Based on this information, which of the following statements is correct?
A) Phosphoric acid is a weaker acid than nitrous acid.
B) Nitrous acid is a weaker acid than water.
C) The nitrite anion is a weaker base than the dihydrogen phosphate anion.
D) The dihydrogen phosphate anion is a stronger acid than nitrous acid.
E) Phosphoric acid is a stronger acid than nitrous acid.
Aqueous solutions of phosphoric acid and sodium nitrite are combined, and the following equilibrium is established. H3PO4(aq) + NO2-(aq) <----> H2PO4-(aq) + HNO2(aq) The equilibrium constant Kc for this reaction is greater than one. Based on this information, which of the following statements is correct?
A) Phosphoric acid is a weaker acid than nitrous acid.
B) Nitrous acid is a weaker acid than water.
C) The nitrite anion is a weaker base than the dihydrogen phosphate anion.
D) The dihydrogen phosphate anion is a stronger acid than nitrous acid.
E) Phosphoric acid is a stronger acid than nitrous acid.
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Please arrange the following acids in order of increasing strength.
| H3PO4 < H2PO4- < HPO4 ^-2 |
| H2PO4- < H3PO4 < HPO4 ^-2 |
| HPO4-2 < H2PO4- < H3PO4 |
| H3PO4 < HPO4 ^-2 < H2PO4- |
| All of these acids have the same strength |
Consider the following Ka values;
acetic acid Ka[CH3COOH (aq)] = 1.80 x 10^-5
chloroacetic acid Ka[CH2ClCOOH (aq)] = 1.40 x 10^-3
hypochlorous acid Ka[HClO (aq)] = 2.90 x 10^-8
hydrofluoric acid Ka[HF (aq)] = 6.30 x 10^-4
formic acid Ka[HCOOH (aq)] = 1.80 x 10^-4
Which of the following is a correct statement?
| Hypochlorous acid is the weakest acid listed |
| Acetic acid is a weaker acid than hypochlorous acid |
| Chloroacetic acid is the strongest acid on the list |
| Formic acid is a stronger acid than acetic acid |
| Formic acid is a weaker acid than hydrofluoric acid |
Which of the following statements strong and weak acids is correct?
| Weak acids completely dissociate in solution |
| Strong acids completely dissociate in solution |
| A 0.05 M solution of an acid is a weak acid |
| Strong acids only partially dissociate in solution |
| Weak acids completely dissociate in solution |
Which of the following is not an Arrhenius acid, but is a Lewis acid??
| HSO4- |
| AlCl3 |
| CH3COOH |
| HCl |
| H2O |
Which of the following compounds is not amphoteric?
| H2O |
| H2CO3 |
| HSO4- |
| HCO3- |
| H2PO4- |
Calculate the pH change that results when 12 mL of 5.9 M NaOH is added to 763 mL of each the following solutions. (See the appendix.)
(a) pure water
(b) 0.10 M NH4Cl
(c) 0.10 M NH3
(d) a solution that is 0.10 M in each NH4+ and NH3
| Acid Name | ConjugateAcid | Ka | pKa | Conjugate Base | Base Name |
|---|---|---|---|---|---|
| perchloric acid | HClO4 | >>1 | < 0 | ClO41- | perchlorate ion |
| hydrohalic acid | HX (X=I,Br,Cl) | >>1 | < 0 | X1- | halide ion |
| sulfuric acid | H2SO4 | >>1 | < 0 | HSO41- | hydrogen sulfate ion |
| nitric acid | HNO3 | >>1 | < 0 | NO31- | nitrate ion |
| hydronium ion | H3O1+ | 1.0 | 0.00 | H2O | water |
| iodic acid | HIO3 | 0.17 | 0.77 | IO31- | iodate ion |
| oxalic acid | H2C2O4 | 5.9 x 10-2 | 1.23 | HC2O41- | hydrogen oxalate ion |
| sulfurous acid | H2SO3 | 1.5 x 10-2 | 1.82 | HSO31- | hydrogen sulfite ion |
| hydrogen sulfate ion | HSO41- | 1.2 x 10-2 | 1.92 | SO42- | sulfate ion |
| phosphoric acid | H3PO4 | 7.5 x 10-3 | 2.12 | H2PO41- | dihydrogen phosphate ion |
| hydrofluoric acid | HF | 7.2 x 10-4 | 3.14 | F1- | fluoride ion |
| nitrous acid | HNO2 | 4.0 x 10-4 | 3.40 | NO21- | nitrite ion |
| lactic acid | HC3H5O3 | 6.4 x 10-5 | 3.85 | C3H5O31- | lactate ion |
| formic acid | HCHO2 | 1.8 x 10-4 | 3.74 | CHO21- | formate ion |
| hydrogen oxalate ion | HC2O41- | 6.4 x 10-5 | 4.19 | C2O42- | oxalate ion |
| hydrazoic acid | HN3 | 1.9 x 10-5 | 4.72 | N31- | azide ion |
| acetic acid | HC2H3O2 | 1.8 x 10-5 | 4.74 | C2H3O21- | acetate ion |
| carbonic acid | H2CO3 | 4.3 x 10-7 | 6.37 | HCO31- | hydrogen carbonate ion |
| hydrogen sulfite ion | HSO31- | 1.0 x 10-7 | 7.00 | SO32- | sulfite ion |
| hydrosulfuric acid | H2S | 1.0 x 10-7 | 7.00 | HS1- | hydrogen sulfide ion |
| dihydrogen phosphate ion | H2PO41- | 6.2 x 10-8 | 7.21 | HPO42- | hydrogen phosphate ion |
| hypochlorous acid | HClO | 3.5 x 10-8 | 7.46 | ClO1- | hypochlorite ion |
| ammonium ion | NH41+ | 5.6 x 10-10 | 9.25 | NH3 | ammonia |
| hydrocyanic acid | HCN | 4.0 x 10-10 | 9.40 | CN1- | cyanide ion |
| hydrogen carbonate ion | HCO31- | 4.7 x 10-11 | 10.33 | CO32- | carbonate ion |
| hydrogen phosphate ion | HPO42- | 4.8 x 10-13 | 12.32 | PO43- | phosphate ion |
| hydrogen sulfide ion | HS1- | 1.3 x 10-13 | 12.89 | S2- | sulfide ion |
| water | H2O | 1.0 x 10-14 | 14.00 | OH1- | hydroxide ion |
| ammonia | NH3 | <<10-14 | NH21- | amide ion | |
| hydroxide ion | OH1- | <<10-14 | O2- | oxide ion | |