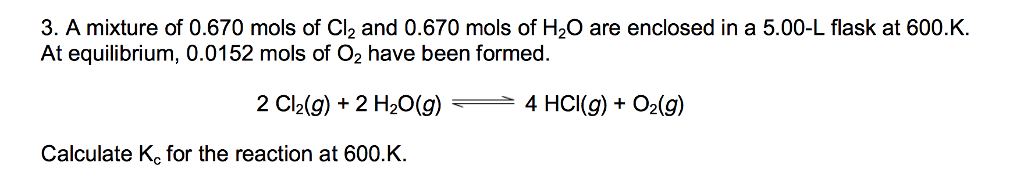1
answer
0
watching
208
views
13 Dec 2019
1)If the temperature is increased and some oxygen is removed, how does this affect the amount of the Cl2 present at equilibrium for the following reaction?
4 HCl(g) + O2(g) ? 2 H2O(g) + 2 Cl2(g) ?H
1)If the temperature is increased and some oxygen is removed, how does this affect the amount of the Cl2 present at equilibrium for the following reaction?
4 HCl(g) + O2(g) ? 2 H2O(g) + 2 Cl2(g) ?H
Casey DurganLv2
17 Dec 2019