1
answer
0
watching
1,281
views
13 Dec 2019
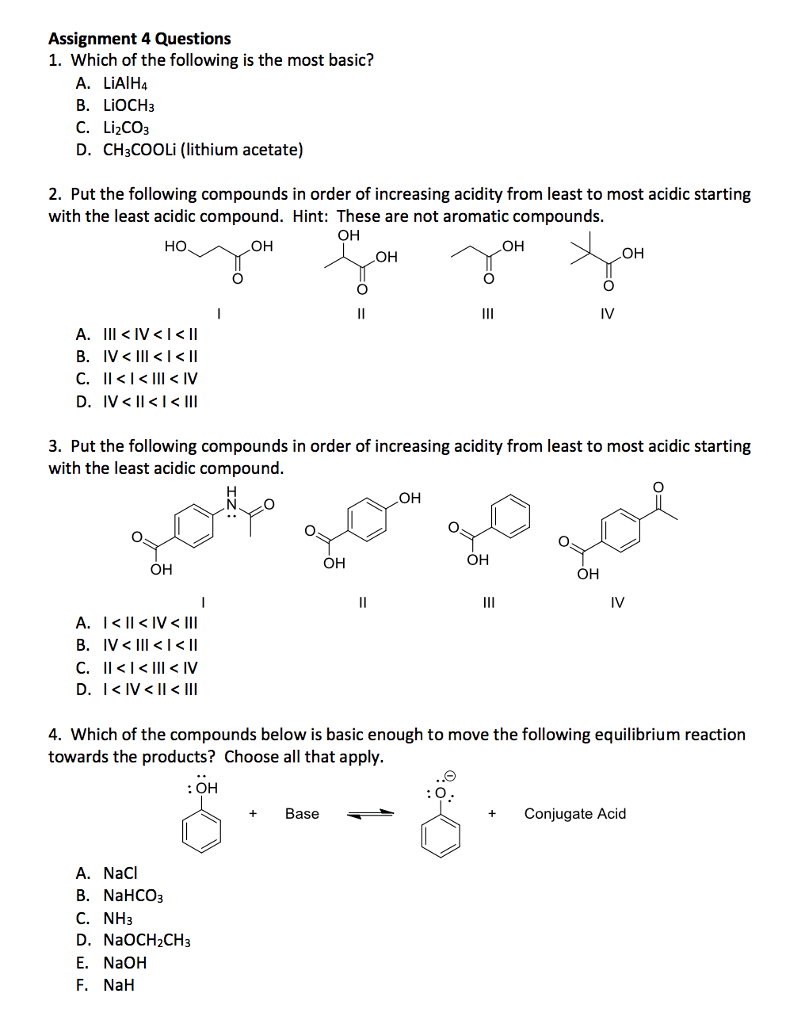
#1 A) Are the solutions made from these compounds acidic, basic or neutral solutions?
a. NaF b. RbCl c. Sn(NO3) d. MgS e. CrI3
#1 B) Acid/base strength
a. Put these in order of increasing basicity
CO3-2, I-, NH2-, HSO4-, OH-, NO3-
b. Put these in order of increasing acidity
HI, NH3, HCO3-, HF, H2O, HClO4
#1 A) Are the solutions made from these compounds acidic, basic or neutral solutions?
a. NaF b. RbCl c. Sn(NO3) d. MgS e. CrI3
#1 B) Acid/base strength
a. Put these in order of increasing basicity
CO3-2, I-, NH2-, HSO4-, OH-, NO3-
b. Put these in order of increasing acidity
HI, NH3, HCO3-, HF, H2O, HClO4
1
answer
0
watching
1,281
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Elin HesselLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232