2
answers
0
watching
366
views
13 Dec 2019
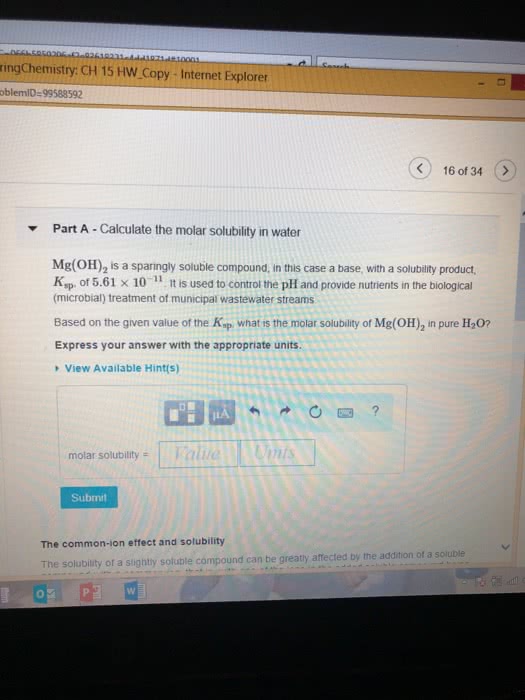
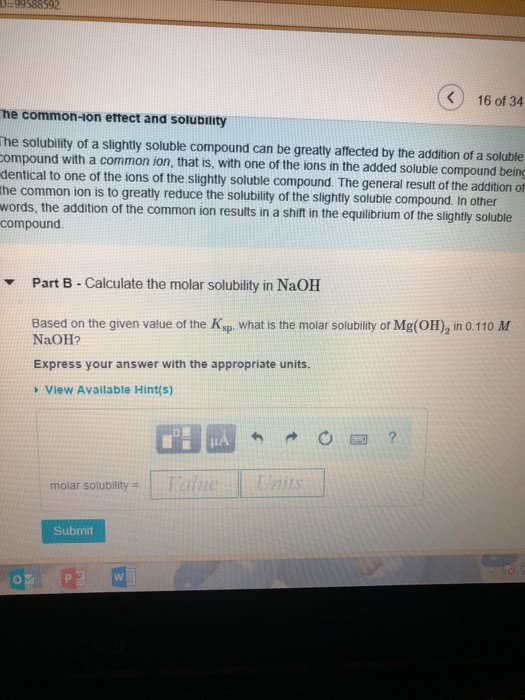
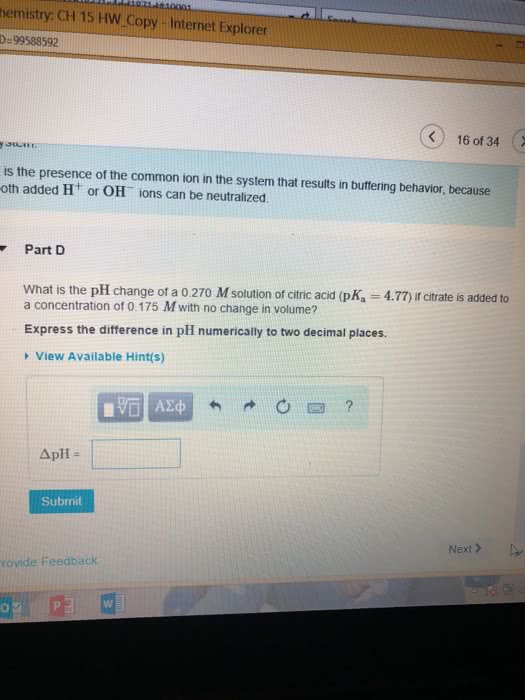
Calculate the molar solubility in waterPart complete Mg(OH)2 is a sparingly soluble compound, in this case a base, with a solubility product, Ksp, of 5.61Ã10?11. It is used to control the pH and provide nutrients in the biological (microbial) treatment of municipal wastewater streams. Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in pure H2O? ANSWER=2.41 x 10^-4 Calculate the molar solubility in NaOH Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in 0.150 M NaOH?
Calculate the molar solubility in waterPart complete Mg(OH)2 is a sparingly soluble compound, in this case a base, with a solubility product, Ksp, of 5.61Ã10?11. It is used to control the pH and provide nutrients in the biological (microbial) treatment of municipal wastewater streams. Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in pure H2O? ANSWER=2.41 x 10^-4 Calculate the molar solubility in NaOH Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in 0.150 M NaOH?
9 Jun 2023
Patrina SchowalterLv2
17 Dec 2019
Already have an account? Log in