1.) average number of moles of NaOH dispensed
2.)average concentration of the acid (mol/L)
3,) Based on your coarse titration volume, do you expect the acetic acid solution to have a higher or lower concentration than the NaOH solution?
higher could you please tell me if number 3 is right
please help with these 2 questions
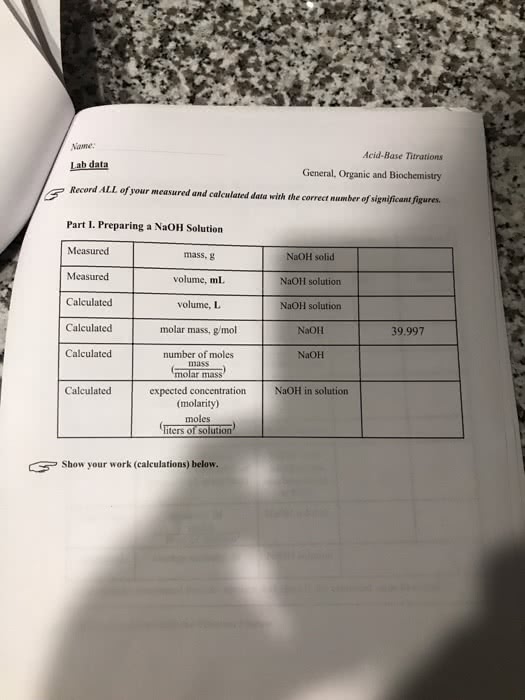
Part 1: Prepare the NaOH Solution
Lab Results
How many mL of water were used to prepare the NaOH solution?
200mL
Data Analysis
Calculate the molarity of the NaOH solution. The molar mass of NaOH is 39.997 g/mol.
0.1M
Calculate the amount of benzoic acid to be neutralized by 20.00 mL NaOH solution, in both moles and grams. The molar mass of benzoic acid is 122.12 g/mol.
1.207 g
Part 2: Perform a Coarse Titration
Lab Results
Record the following data from your course titration in the table below.
mass of benzoic acid used (g) 0.26614g pH of benzoic acid solution before titration 2.96 volume of NaOH in the burette before titration (mL) 50.0mL volume of NaOH in the burette after titration (mL) 28.89mL volume of NaOH dispensed in the titration (mL) 21.11mL
Data Analysis
How do you expect your coarse titration volume to compare to your fine titration volumes?
I expect the values to be a little off from each other since in the first titration we let out a lot at a time and didnt slowly do the titration. should only be off by a few mL.
Part 3: Perform Fine Titrations
Lab Results
Record the volume of NaOH solution dispensed in the 3 fine titrations.
20 mL dispenced ,20.34mL, 20.30mL
Data Analysis
Calculate the average concentration of the NaOH solution, using the average volume of NaOH solution dispensed in the 3 fine titrations. Report your answer using enough significant figures to distinguish it from the expected concentration of 0.100 M.
V = (V1 + V2 + V3) / 3
V = (20.03 mL + 20.06 mL + 20.02 mL) / 3 = 20.04 mL
M = n / V = 0.00200 moles / 0.02004 L = 0.0998 M
Learning objective: Determine the concentration of a solution.
20.21= ----> 0.0999M
Experiment 2: Use the Standardized NaOH Solution to Determine the Concentration of an Acid
Part 1: Perform a Coarse Titration
Lab Results
What was the pH at the end point of the coarse titration?
3.87ph
Data Analysis
Based on your coarse titration volume, do you expect the acetic acid solution to have a higher or lower concentration than the NaOH solution?
higher
Part 2: Perform Fine Titrations
Lab Results
For the 3 fine titrations of the acid of unknown concentration, fill in the following data.
Titration #1 Titration #2 Titration #3 volume of acid (mL) 59.11 40.97 40.98 volume of NaOH dispensed (mL) 9.05 9.03 9.02
Data Analysis
Calculate the following quantities and record the data in the table below.
average volume of NaOH solution dispensed (mL) 9.03 average number of moles of NaOH dispensed average concentration of the acid (mol/L)
Conclusions
Phenolphthalein is pink over the range of pH 8 â 12. Why was it a useful indicator of when the equivalence point was reached?
This was helpful because once I got to the ph of 7 I knew that I would be getting closer to the end point which would change the color to pink.
Suppose a student titrated a sample of monoprotic acid of unknown concentration using a previously standardized solution of NaOH.
volume of 0.125 M NaOH dispensed 24.68 mL volume of acid solution 50.00 mL
Given the data in the figure above, what is the concentration of the unknown acid?
0.0617M
Lab NOTES:
ex1
Take sodium hydroxide from the Materials shelf and add 0.800 g to the beaker.
Take water from the Materials shelf and add 200 mL to the beaker.
Ph 2.96
inital volume in burett
inital volume 50ml
1.) volume43.78mL
volumes dispenced =6.22ml
2. volume =38.05
volume dispenced =11.95ml
3.)volume = 32.06
volume dispenced=17.94
4.28.89ml
volume dispenced=21.11ml
ph=10.95
--------------------------------------
Ex 2
inital ph= 2.96
volume30.33
dispence 20.05
1. volume 39.92ml
volume dispenced=10.08
2.volume 29.66
voiume dispence 20.34
final ph10.43
initial valume in burett 50ml
--------------------
ex3
final ph9.43
volume 30.28
volume dipenced=20.00
----------------------------------------------------------------------
2nd Ex
initial ph 3.11
volume 50
1. volume 48.95mL
dispenced 1.05mL
ph 3.87
2. volume 45.88
dispenced 4.12
ph 4.67
3 volume 42.99
dispenced 7.01
ph 5.29
4 volume 39.93
dispence 10.07
ph 11.45
--------------------
F1
final ph 9.99
volume 59.11
dispenced 9.05
F2
ph 9.59
volume 40.97
dispenced 9.03
f3
ph 9.50
volume 40.98
9.02
| 1.) average number of moles of NaOH dispensed |
|---|
| 2.)average concentration of the acid (mol/L) |
|---|
3,) Based on your coarse titration volume, do you expect the acetic acid solution to have a higher or lower concentration than the NaOH solution?
| higher could you please tell me if number 3 is right |
please help with these 2 questions
Part 1: Prepare the NaOH Solution
Lab Results
How many mL of water were used to prepare the NaOH solution?
| 200mL |
Data Analysis
Calculate the molarity of the NaOH solution. The molar mass of NaOH is 39.997 g/mol.
| 0.1M |
Calculate the amount of benzoic acid to be neutralized by 20.00 mL NaOH solution, in both moles and grams. The molar mass of benzoic acid is 122.12 g/mol.
| 1.207 g |
Part 2: Perform a Coarse Titration
Lab Results
Record the following data from your course titration in the table below.
| mass of benzoic acid used (g) | 0.26614g |
|---|---|
| pH of benzoic acid solution before titration | 2.96 |
| volume of NaOH in the burette before titration (mL) | 50.0mL |
| volume of NaOH in the burette after titration (mL) | 28.89mL |
| volume of NaOH dispensed in the titration (mL) | 21.11mL |
Data Analysis
How do you expect your coarse titration volume to compare to your fine titration volumes?
| I expect the values to be a little off from each other since in the first titration we let out a lot at a time and didnt slowly do the titration. should only be off by a few mL. |
Part 3: Perform Fine Titrations
Lab Results
Record the volume of NaOH solution dispensed in the 3 fine titrations.
| 20 mL dispenced ,20.34mL, 20.30mL |
Data Analysis
Calculate the average concentration of the NaOH solution, using the average volume of NaOH solution dispensed in the 3 fine titrations. Report your answer using enough significant figures to distinguish it from the expected concentration of 0.100 M.
| V = (V1 + V2 + V3) / 3 V = (20.03 mL + 20.06 mL + 20.02 mL) / 3 = 20.04 mL M = n / V = 0.00200 moles / 0.02004 L = 0.0998 M Learning objective: Determine the concentration of a solution. 20.21= ----> 0.0999M |
Experiment 2: Use the Standardized NaOH Solution to Determine the Concentration of an Acid
Part 1: Perform a Coarse Titration
Lab Results
What was the pH at the end point of the coarse titration?
| 3.87ph |
Data Analysis
Based on your coarse titration volume, do you expect the acetic acid solution to have a higher or lower concentration than the NaOH solution?
| higher |
Part 2: Perform Fine Titrations
Lab Results
For the 3 fine titrations of the acid of unknown concentration, fill in the following data.
| Titration #1 | Titration #2 | Titration #3 | |
|---|---|---|---|
| volume of acid (mL) | 59.11 | 40.97 | 40.98 |
| volume of NaOH dispensed (mL) | 9.05 | 9.03 | 9.02 |
Data Analysis
Calculate the following quantities and record the data in the table below.
| average volume of NaOH solution dispensed (mL) | 9.03 |
|---|---|
| average number of moles of NaOH dispensed | |
| average concentration of the acid (mol/L) |
Conclusions
Phenolphthalein is pink over the range of pH 8 â 12. Why was it a useful indicator of when the equivalence point was reached?
| This was helpful because once I got to the ph of 7 I knew that I would be getting closer to the end point which would change the color to pink. |
Suppose a student titrated a sample of monoprotic acid of unknown concentration using a previously standardized solution of NaOH.
| volume of 0.125 M NaOH dispensed | 24.68 mL |
|---|---|
| volume of acid solution | 50.00 mL |
Given the data in the figure above, what is the concentration of the unknown acid?
| 0.0617M Lab NOTES: ex1 Take sodium hydroxide from the Materials shelf and add 0.800 g to the beaker. Take water from the Materials shelf and add 200 mL to the beaker. Ph 2.96 inital volume in burett inital volume 50ml 1.) volume43.78mL volumes dispenced =6.22ml 2. volume =38.05 volume dispenced =11.95ml 3.)volume = 32.06 volume dispenced=17.94 4.28.89ml volume dispenced=21.11ml ph=10.95 -------------------------------------- Ex 2 inital ph= 2.96 volume30.33 dispence 20.05 1. volume 39.92ml volume dispenced=10.08 2.volume 29.66 voiume dispence 20.34 final ph10.43 initial valume in burett 50ml -------------------- ex3 final ph9.43 volume 30.28 volume dipenced=20.00 ---------------------------------------------------------------------- 2nd Ex initial ph 3.11 volume 50 1. volume 48.95mL dispenced 1.05mL ph 3.87 2. volume 45.88 dispenced 4.12 ph 4.67 3 volume 42.99 dispenced 7.01 ph 5.29 4 volume 39.93 dispence 10.07 ph 11.45 -------------------- F1 final ph 9.99 volume 59.11 dispenced 9.05 F2 ph 9.59 volume 40.97 dispenced 9.03 f3 ph 9.50 volume 40.98 9.02 |