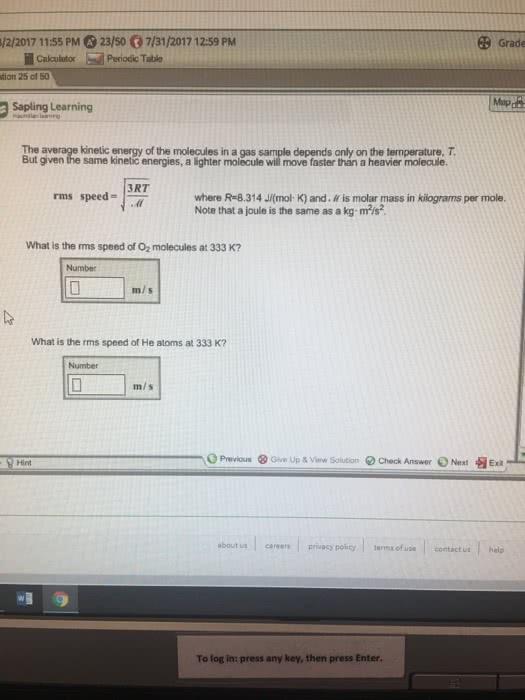
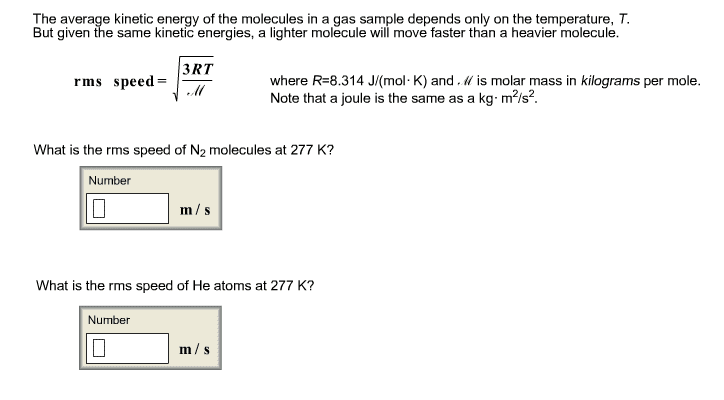
1) 1.75g of an unknown gas at 31°C and 1.05 atm is stores in a 2.55-L flask.
- What is the density of the gas?
______ g/L
- What is the molar mass of the gas?
_____ g/mol
2) If the absolute temperature of a gas is tripled, what happens to the root- mean- square speed of the molecules? (nothing,the new rms speed is 9 times the original rms speed, the new rms speed is 3 times the original rms speed, the new rms speed is 1.732 times the original rms speed, the new rms speed is 1/3 the orginal rms speed)
3) Calculate the average translational kinetic energy (sometimes just called average kinetic energy)...
- for one mole of gas at 723 K.
Ek= ________ J/mol
- for a single gas molecule at 723 K
Ek= ________ J/molecule
4) If He(g) has an average kinetic ebergy of 8890 J/mol inder certain conditions, what is the root mean square speed of F2(g) molecules under the same conditions?
- ___________ m/s
5) Use the van der Waals equation os state to calculate the pressure of 2.70 mol of H2O at 477 K in a 5.10-L vessel.
- P= _________ atm
- use the ideal gas equation to calculate the oressure under the same conditions.
P= _______ atm
1) 1.75g of an unknown gas at 31°C and 1.05 atm is stores in a 2.55-L flask.
- What is the density of the gas?
______ g/L
- What is the molar mass of the gas?
_____ g/mol
2) If the absolute temperature of a gas is tripled, what happens to the root- mean- square speed of the molecules? (nothing,the new rms speed is 9 times the original rms speed, the new rms speed is 3 times the original rms speed, the new rms speed is 1.732 times the original rms speed, the new rms speed is 1/3 the orginal rms speed)
3) Calculate the average translational kinetic energy (sometimes just called average kinetic energy)...
- for one mole of gas at 723 K.
Ek= ________ J/mol
- for a single gas molecule at 723 K
Ek= ________ J/molecule
4) If He(g) has an average kinetic ebergy of 8890 J/mol inder certain conditions, what is the root mean square speed of F2(g) molecules under the same conditions?
- ___________ m/s
5) Use the van der Waals equation os state to calculate the pressure of 2.70 mol of H2O at 477 K in a 5.10-L vessel.
- P= _________ atm
- use the ideal gas equation to calculate the oressure under the same conditions.
P= _______ atm