1
answer
0
watching
1,646
views
13 Dec 2019
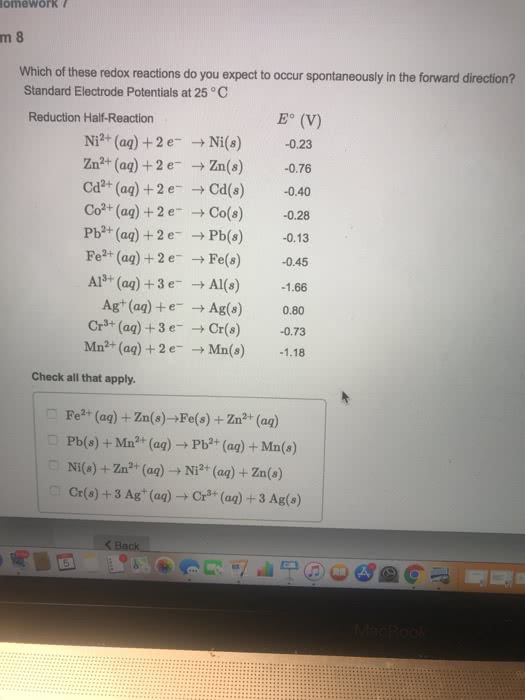
Which of the following redox reactions do you expect to occur spontaneously in the reverse direction? (Hint: The reactions are occurring under standard conditions (1 M for the aqueous ions).)
```2Al(s)+3Pb2+(aq) ? 2Al3+(aq)+3Pb(s) Fe(s)+Cr3+(aq) ? Fe3+(aq)+Cr(s) 2Cu+(aq)+Co(s) ? 2Cu(s)+Co2+(s) Ca2+(aq)+Zn(s) ? Ca(s)+Zn2+(aq)
please help, I know that A reaction with E?cell greater than zero will not occur spontaneously in the reverse direction. Recall that a reaction will occur spontaneously in the reverse direction when the E?cell of a reaction is less than zero.
Which of the following redox reactions do you expect to occur spontaneously in the reverse direction? (Hint: The reactions are occurring under standard conditions (1 M for the aqueous ions).)
| ```2Al(s)+3Pb2+(aq) ? 2Al3+(aq)+3Pb(s) | |
| Fe(s)+Cr3+(aq) ? Fe3+(aq)+Cr(s) | |
| 2Cu+(aq)+Co(s) ? 2Cu(s)+Co2+(s) | |
| Ca2+(aq)+Zn(s) ? Ca(s)+Zn2+(aq) |
please help, I know that A reaction with E?cell greater than zero will not occur spontaneously in the reverse direction. Recall that a reaction will occur spontaneously in the reverse direction when the E?cell of a reaction is less than zero.
Nestor RutherfordLv2
17 Dec 2019