1
answer
0
watching
293
views
13 Dec 2019
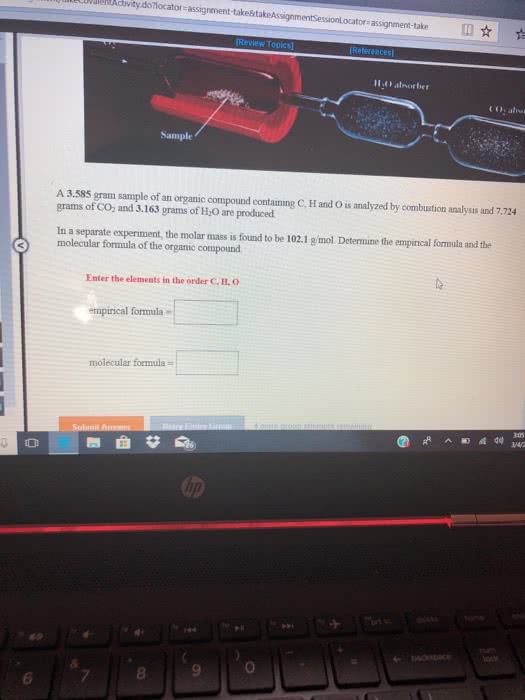
A 16.43 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis and 16.06 grams of CO2 and 3.288 grams of H2O are produced.
In a separate experiment, the molar mass is found to be 90.04 g/mol. Determine the empirical formula and the molecular formula of the organic compound.
Enter the elements in the order C, H, O
empirical formula =
molecular formula =
A 16.43 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis and 16.06 grams of CO2 and 3.288 grams of H2O are produced.
In a separate experiment, the molar mass is found to be 90.04 g/mol. Determine the empirical formula and the molecular formula of the organic compound.
Enter the elements in the order C, H, O
| empirical formula = |
| molecular formula = |
Beverley SmithLv2
17 Dec 2019