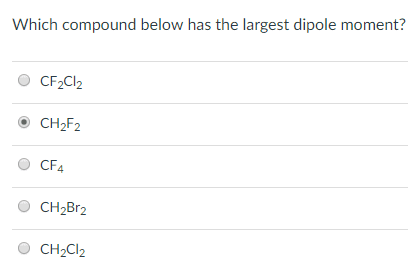1
answer
0
watching
7,832
views
13 Dec 2019
Which compound (CH2F2 or CF4) experiences stronger intermolecular attractive forces overall?
Which compound (CH2F2 or CF4) experiences stronger intermolecular attractive forces overall?
Verified Answer
Sixta KovacekLv2
17 Dec 2019
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University5 May 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.