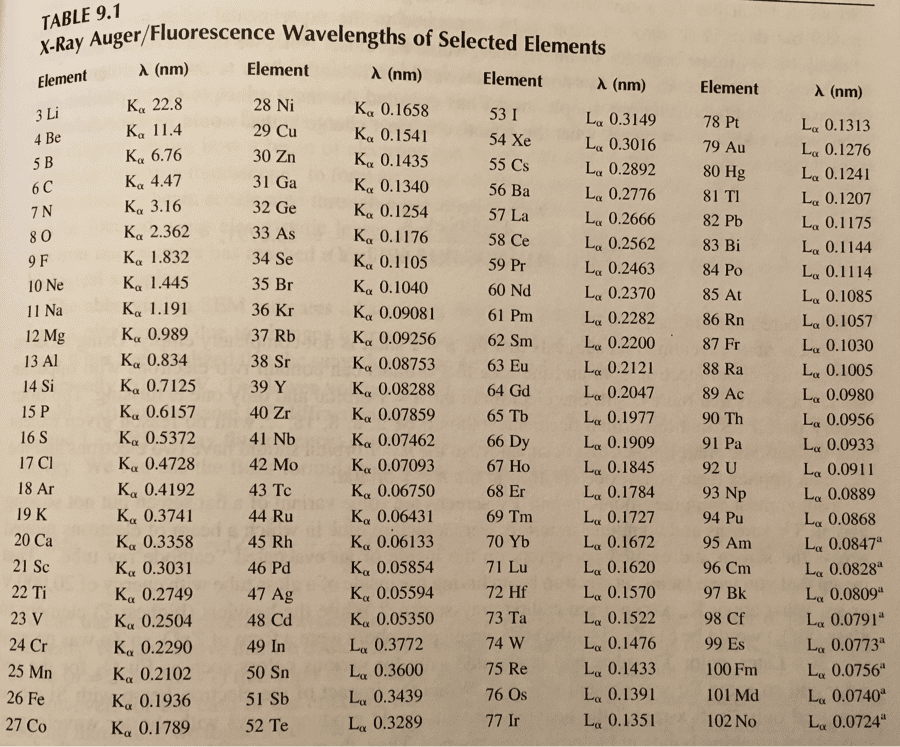
Please help complete the table. Based on the experiment that was conducted. The unknown metal was placed in 40 mL of water.
Trial 1: Time (minutes)
Trial 1:
Temp (°C)
Trial 2: Time (minutes)
Trial 2:
Temp (°C)
Trial 3: Time (minutes)
Trial 3:
Temp (°C)
Metal: Initial
102
Metal: Initial
99
Metal: Initial
98
Calorimeter: initial
32
Calorimeter: initial
34
Calorimeter: initial
29
Calorimeter + Metal: 1 min
36
Calorimeter + Metal: 1 min
39
Calorimeter + Metal: 1 min
31
Calorimeter + Metal: 2 min
42
Calorimeter + Metal: 2 min
40
Calorimeter + Metal: 2 min
36
Calorimeter + Metal: 3 min
45
Calorimeter + Metal: 3 min
42
Calorimeter + Metal: 3 min
39
Calorimeter + Metal: 4 min
46
Calorimeter + Metal: 4 min
44
Calorimeter + Metal: 4 min
41
Calorimeter + Metal: 5 min
48
Calorimeter + Metal: 5 min
46
Calorimeter + Metal: 5 min
43
DT for water
DT for water
DT for water
DT for metal
DT for metal
DT for metal
Mass of water: 40 (g)
Specific Heat capacity of water (J/g°C):
Calculation for Qwater:
Mass of metal (g): 19
Qmetal = - Qwater =
Calculation for Csp of metal (Trial 1):
Calculation for Csp of metal (Trial 2):
Calculation for Csp of metal (Trial 3):
Average Csp of metal:
Identity of Metal (extra credit):
*include evidence/explanation below table
Please help complete the table. Based on the experiment that was conducted. The unknown metal was placed in 40 mL of water.
| Trial 1: Time (minutes) | Trial 1: Temp (°C) | Trial 2: Time (minutes) | Trial 2: Temp (°C) | Trial 3: Time (minutes) | Trial 3: Temp (°C) | ||||
| Metal: Initial | 102 | Metal: Initial | 99 | Metal: Initial | 98 | ||||
| Calorimeter: initial | 32 | Calorimeter: initial | 34 | Calorimeter: initial | 29 | ||||
| Calorimeter + Metal: 1 min | 36 | Calorimeter + Metal: 1 min | 39 | Calorimeter + Metal: 1 min | 31 | ||||
| Calorimeter + Metal: 2 min | 42 | Calorimeter + Metal: 2 min | 40 | Calorimeter + Metal: 2 min | 36 | ||||
| Calorimeter + Metal: 3 min | 45 | Calorimeter + Metal: 3 min | 42 | Calorimeter + Metal: 3 min | 39 | ||||
| Calorimeter + Metal: 4 min | 46 | Calorimeter + Metal: 4 min | 44 | Calorimeter + Metal: 4 min | 41 | ||||
| Calorimeter + Metal: 5 min | 48 | Calorimeter + Metal: 5 min | 46 | Calorimeter + Metal: 5 min | 43 | ||||
| DT for water | DT for water | DT for water | |||||||
| DT for metal | DT for metal | DT for metal | |||||||
| Mass of water: 40 (g) | Specific Heat capacity of water (J/g°C): | ||||||||
| Calculation for Qwater: | |||||||||
| Mass of metal (g): 19 | Qmetal = - Qwater = | ||||||||
| Calculation for Csp of metal (Trial 2): | Calculation for Csp of metal (Trial 3): | |||||||
| Average Csp of metal: | Identity of Metal (extra credit): *include evidence/explanation below table | ||||||||