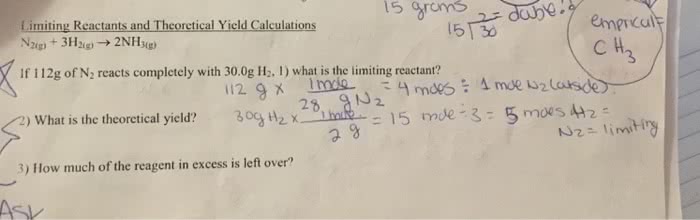1
answer
0
watching
284
views
13 Dec 2019
Answer the following questions using the balanced chemical equation below:
Bi2O3 + 3 C ? 2 Bi + 3 CO
a. What is the limiting reactant when 232.47 g Bi2O3 reacts with 27.03 g C?
b. What is the mass of bismuth produced from this reaction (i.e. the theoretical yield)?
c. If the actual yield is 123.20 g Bi, what is the percent yield?
d. How much excess reagent is left?
Answer the following questions using the balanced chemical equation below:
Bi2O3 + 3 C ? 2 Bi + 3 CO
a. What is the limiting reactant when 232.47 g Bi2O3 reacts with 27.03 g C?
b. What is the mass of bismuth produced from this reaction (i.e. the theoretical yield)?
c. If the actual yield is 123.20 g Bi, what is the percent yield?
d. How much excess reagent is left?
Hubert KochLv2
17 Dec 2019