1
answer
0
watching
909
views
13 Dec 2019
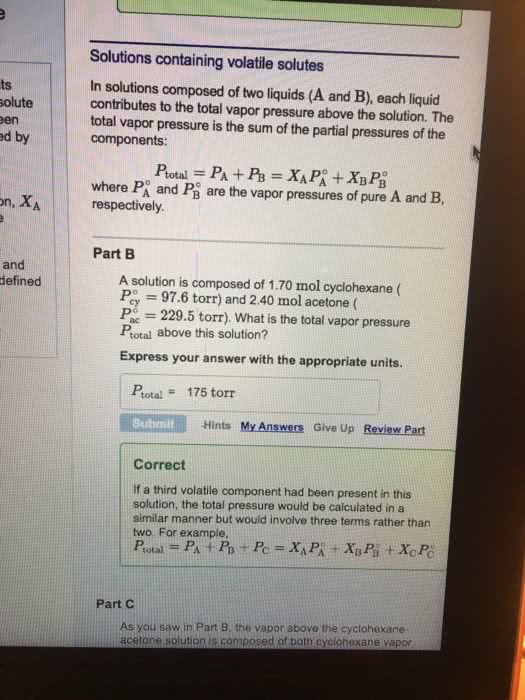
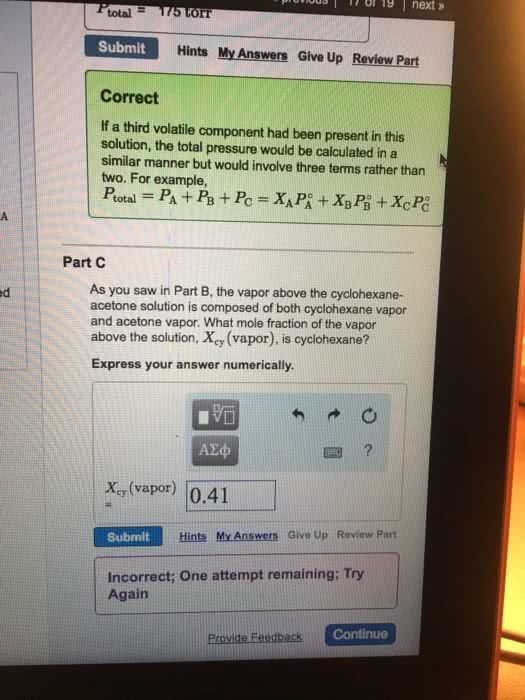
Which of the following statements are true about an ideal solution of two volatile liquids?
A. The partial pressure of each component above the liquid is given by Raoult's law.
B.The liquid and gaseous phases can only coexist for a single value of the pressure at a given temperature.
C. An ideal solution of two volatile liquids can exist over a range of pressures that are limited by the pressure for which only a trace of liquid remains, and the pressure for which only a trace of gas remains.
D. The vapor phase is always depleted in the more volatile or higher vapor pressure component in comparison with the liquid phase.
Answer choices:
A and B
A and C
C and D
B and D
Which of the following statements are true about an ideal solution of two volatile liquids?
A. The partial pressure of each component above the liquid is given by Raoult's law.
B.The liquid and gaseous phases can only coexist for a single value of the pressure at a given temperature.
C. An ideal solution of two volatile liquids can exist over a range of pressures that are limited by the pressure for which only a trace of liquid remains, and the pressure for which only a trace of gas remains.
D. The vapor phase is always depleted in the more volatile or higher vapor pressure component in comparison with the liquid phase.
Answer choices:
A and B
A and C
C and D
B and D
Lelia LubowitzLv2
17 Dec 2019