1
answer
0
watching
140
views
13 Dec 2019
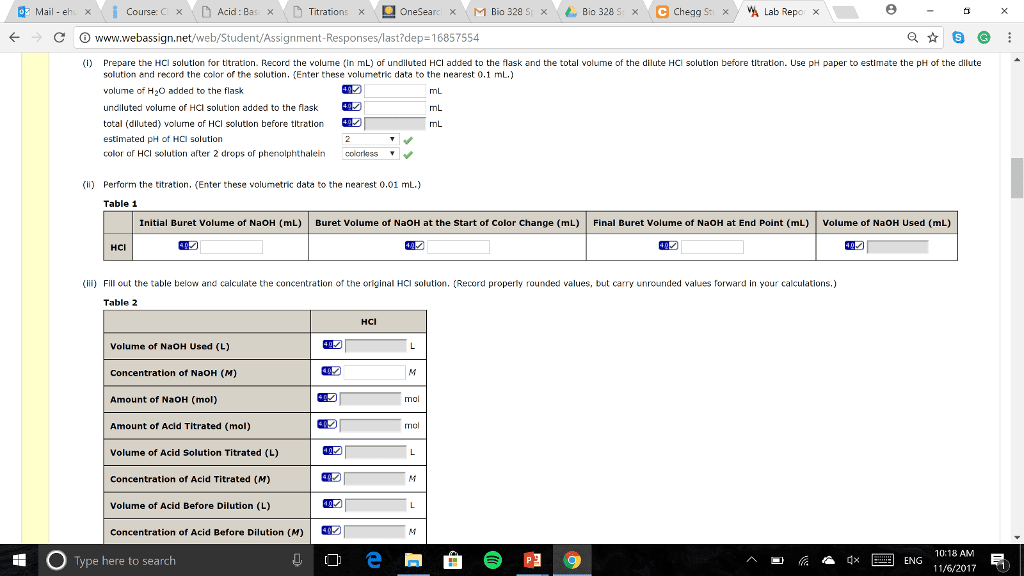
In order to determing the concrentration of an HCl soultion, a student titrated 10.00 mL of HCl solution with 0.1500 M NaOH. After adding 26.18 mL of the NaOH soultion, the student realized he had inadvertenly gone past the end point. The student then added 2.00 mL more of the original HCl solution. The student then completed the titration requiring 1.65 mL more of the NaOH solution. WHat was the molar concentration of the HCl solution
In order to determing the concrentration of an HCl soultion, a student titrated 10.00 mL of HCl solution with 0.1500 M NaOH. After adding 26.18 mL of the NaOH soultion, the student realized he had inadvertenly gone past the end point. The student then added 2.00 mL more of the original HCl solution. The student then completed the titration requiring 1.65 mL more of the NaOH solution. WHat was the molar concentration of the HCl solution
Elin HesselLv2
17 Dec 2019