1
answer
0
watching
44
views
13 Dec 2019
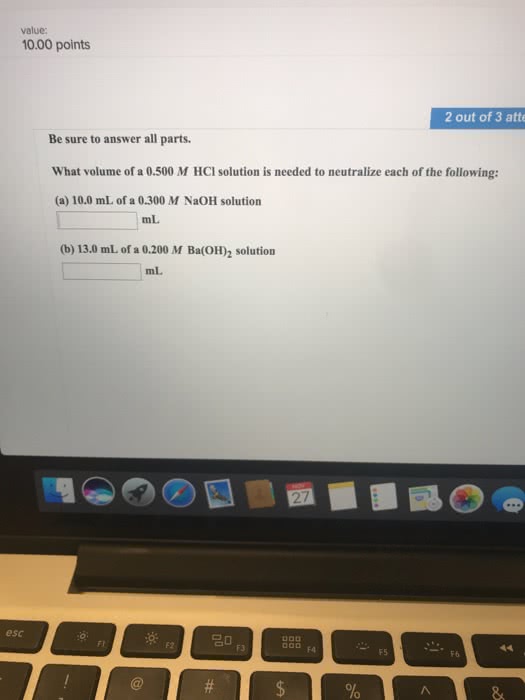
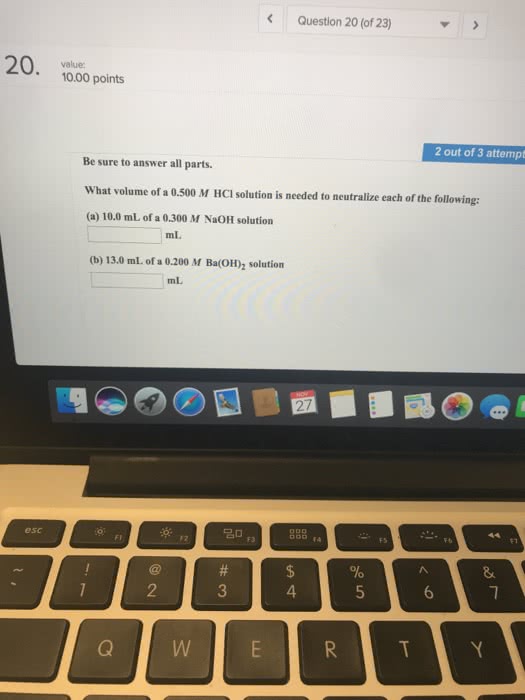
1.what is the volume of 4.029 M NaOH is needed to react with 47.38 ml of 3.1110 M H2SO4?
2. at 25.0°C a 40.00 ml sample of a saturated solution of Ba (OH)2 is neutralized by 29.10 ml of 0.300 M HCl.calculate the Ksp of Ba (OH)2
1.what is the volume of 4.029 M NaOH is needed to react with 47.38 ml of 3.1110 M H2SO4?
2. at 25.0°C a 40.00 ml sample of a saturated solution of Ba (OH)2 is neutralized by 29.10 ml of 0.300 M HCl.calculate the Ksp of Ba (OH)2
Patrina SchowalterLv2
17 Dec 2019