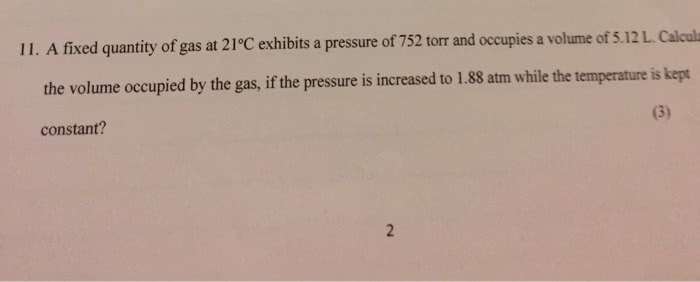1
answer
0
watching
71
views
13 Dec 2019
If a sample of gas occupies a volume of 10.0 L at STP, what will be the volume occupied when the temperature is changed to 47 oC and the pressure to 660 torr?
If a sample of gas occupies a volume of 10.0 L at STP, what will be the volume occupied when the temperature is changed to 47 oC and the pressure to 660 torr?
Lelia LubowitzLv2
17 Dec 2019