1
answer
0
watching
653
views
13 Dec 2019
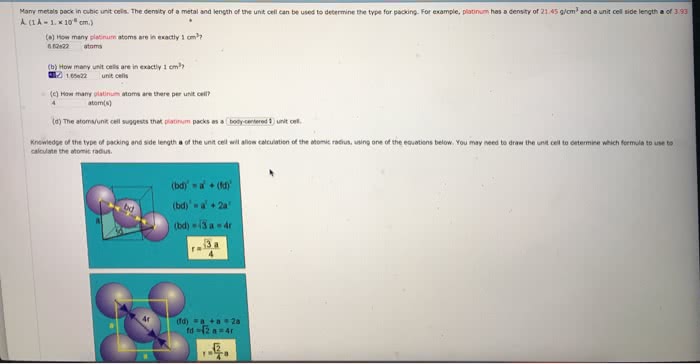
Many metals pack in cubic unit cells. The density of a metal and length of the unit cell can be used to determine the type for packing. For example, sodium has a density of 0.968 g/cm3 and a unit cell side length a of 4.29 Ã . (1 Ã = 1. 10-8 cm.)
(a) How many sodium atoms are in exactly 1 cm3?
(b) How many unit cells are in exactly 1 cm3?
(c) How many sodium atoms are there per unit cell?
The atoms/unit cell suggests that sodium packs as a ( simple body-centered face-centered )unit cell.
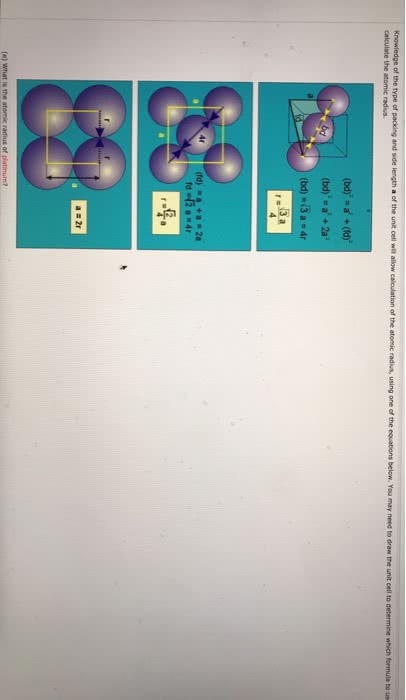
(e) What is the atomic radius of sodium?
Many metals pack in cubic unit cells. The density of a metal and length of the unit cell can be used to determine the type for packing. For example, sodium has a density of 0.968 g/cm3 and a unit cell side length a of 4.29 Ã . (1 Ã = 1. 10-8 cm.)
(a) How many sodium atoms are in exactly 1 cm3?
(b) How many unit cells are in exactly 1 cm3?
(c) How many sodium atoms are there per unit cell?
The atoms/unit cell suggests that sodium packs as a ( simple body-centered face-centered )unit cell.
(e) What is the atomic radius of sodium?
Hubert KochLv2
17 Dec 2019