1
answer
0
watching
168
views
13 Dec 2019
Moles of Zn(NO3)2 in 151.50 g of this substance.Express your answer using four significant figures.
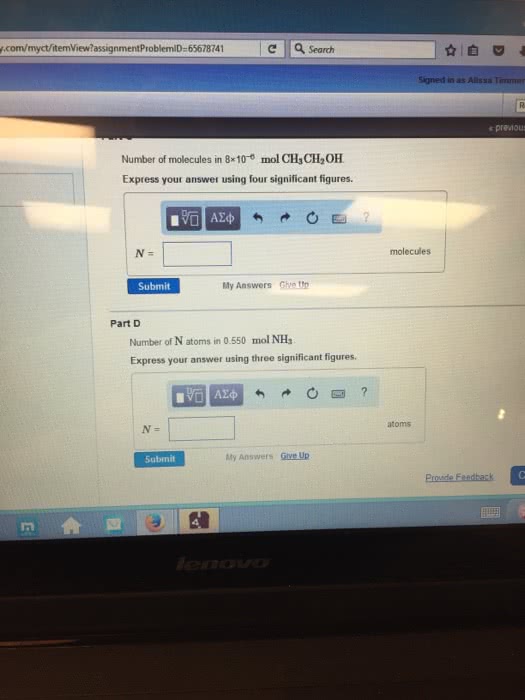
Number of molecules in 6Ã10â6 mol CH3CH2OH. Express your answer using four significant figures.
Number of N atoms in 0.550 mol NH3. Express your answer using three significant figures.
Moles of Zn(NO3)2 in 151.50 g of this substance.Express your answer using four significant figures.
Number of molecules in 6Ã10â6 mol CH3CH2OH. Express your answer using four significant figures.
Number of N atoms in 0.550 mol NH3. Express your answer using three significant figures.
Beverley SmithLv2
17 Dec 2019