1
answer
0
watching
829
views
13 Dec 2019
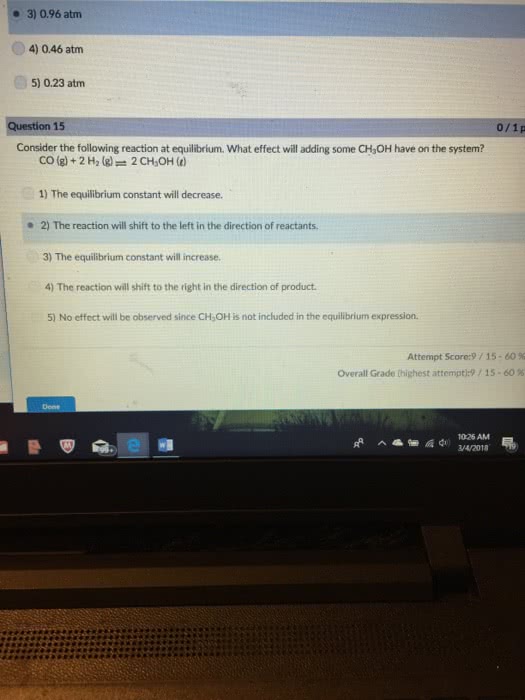
Consider the following reaction at equilibrium. What effect will adding CO2 have on the system?
CO2(g) + C(graphite) â 2 CO(g)
A)The reaction will shift to the right in the direction of products
B)The equilibrium constant will increase
C)The equilibrium constant will decrease
D)The reaction will shift to the left in the direction of reactants
E)No effect will be observed since C is not included in the equilibrium expression
Consider the following reaction at equilibrium. What effect will adding CO2 have on the system?
CO2(g) + C(graphite) â 2 CO(g)
A)The reaction will shift to the right in the direction of products
B)The equilibrium constant will increase
C)The equilibrium constant will decrease
D)The reaction will shift to the left in the direction of reactants
E)No effect will be observed since C is not included in the equilibrium expression
Lelia LubowitzLv2
17 Dec 2019