1
answer
0
watching
264
views
13 Dec 2019
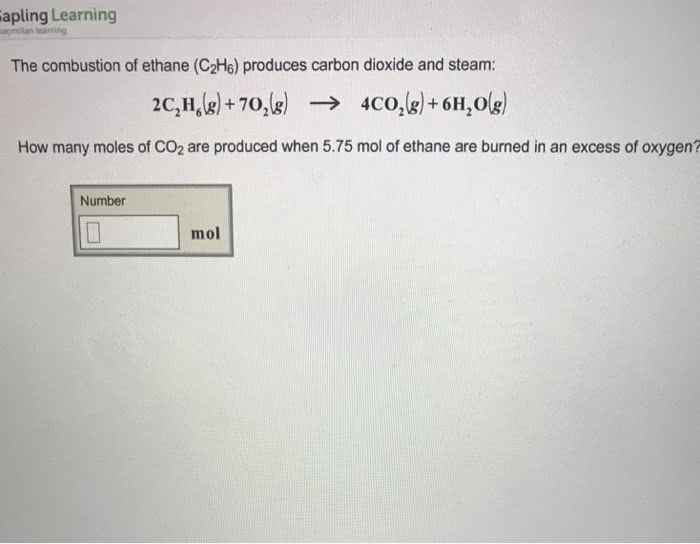
Part A If 1.00 mol of ethane gas and 2.40 mol of oxygen gas react, what is the limiting reactant? 2C2H6(g)+7O2(g)â4CO2(g)+6H2O(g) If 1.00 of ethane gas and 2.40 of oxygen gas react, what is the limiting reactant?
a.) O2
b.) C2H6
c.) neither C2H6 nor O2
Part B
How many moles of water are produced according?
Part A If 1.00 mol of ethane gas and 2.40 mol of oxygen gas react, what is the limiting reactant? 2C2H6(g)+7O2(g)â4CO2(g)+6H2O(g) If 1.00 of ethane gas and 2.40 of oxygen gas react, what is the limiting reactant?
a.) O2
b.) C2H6
c.) neither C2H6 nor O2
Part B
How many moles of water are produced according?
Nelly StrackeLv2
16 Dec 2019