1
answer
0
watching
102
views
13 Dec 2019
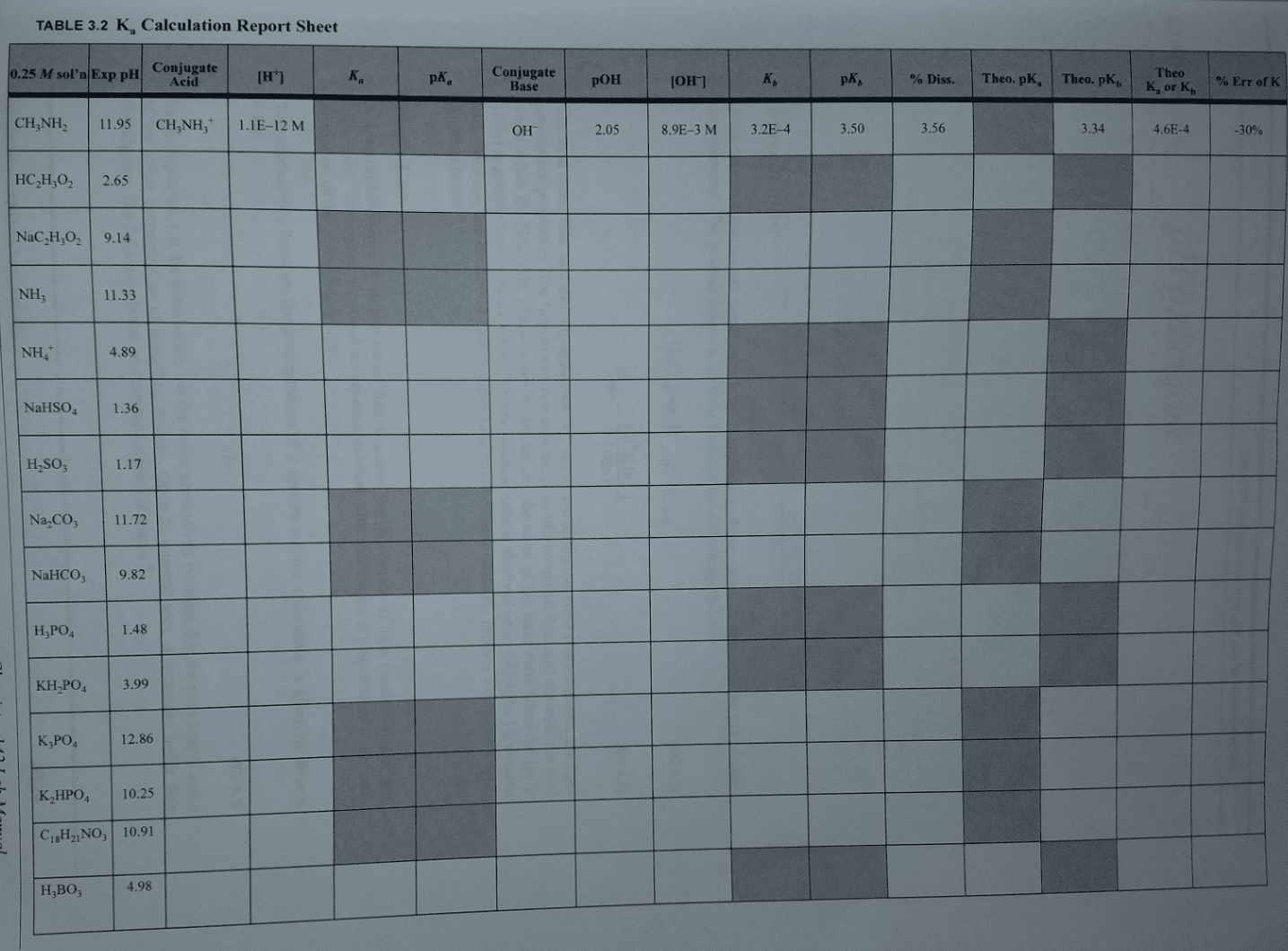
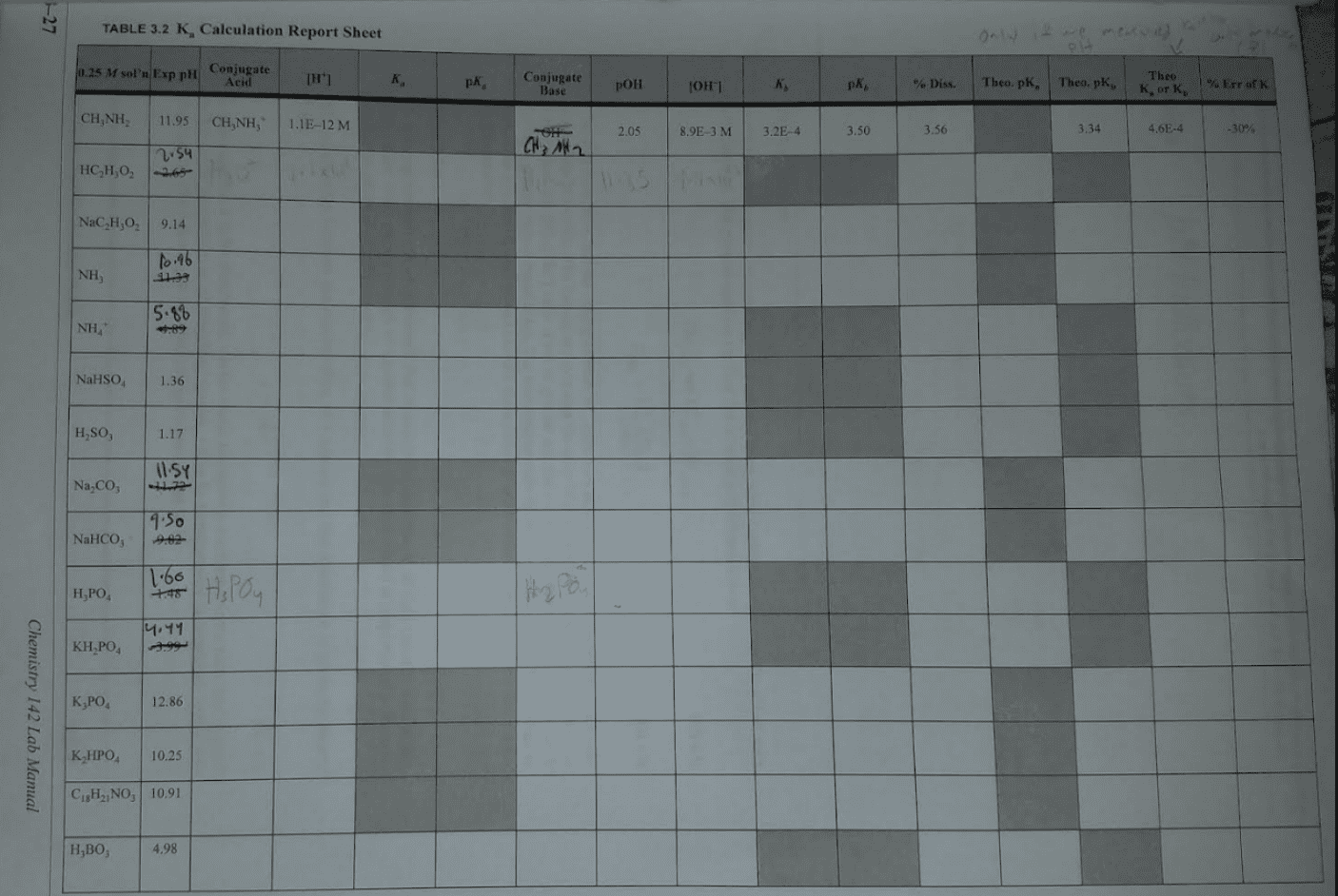
Consider a 300mL sample of 0.250M HCN (Ka=4.9x10^-10). To find the pH, do you need an ice table, can you use the Henderson-Hasselbalch equation, or something else? If an ice table, will it be a Ka or Kb equilibrium? Can you use the small x assumption? Is this a buffer? If something else, then how?
(Need Help Please!!! If possible could you work it out so i can know how to do it)
Consider a 300mL sample of 0.250M HCN (Ka=4.9x10^-10). To find the pH, do you need an ice table, can you use the Henderson-Hasselbalch equation, or something else? If an ice table, will it be a Ka or Kb equilibrium? Can you use the small x assumption? Is this a buffer? If something else, then how?
(Need Help Please!!! If possible could you work it out so i can know how to do it)
1
answer
0
watching
102
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Irving HeathcoteLv2
16 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232