1
answer
0
watching
227
views
12 Dec 2019
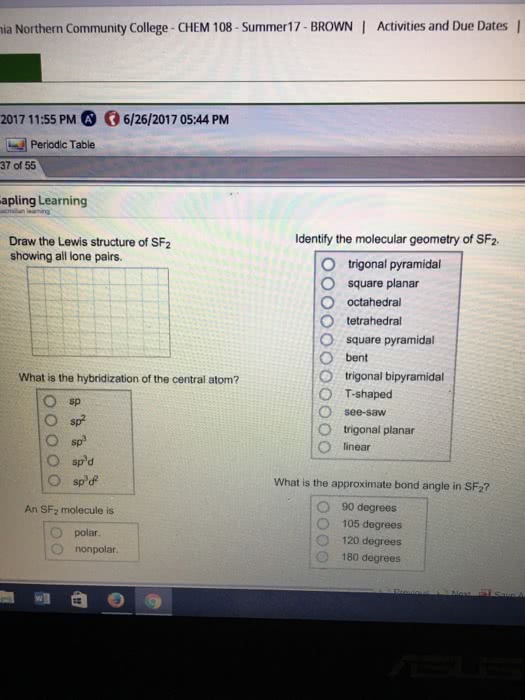
Select all molecular geometries (shapes) that are possible for a molecule with five electron groups an the central atom.
Do not include molecular geometries that can only be produced by having a single bonding group on the central atom (all but one of the electron groups are lone pairs.)
ocahedral T-shaped linear bent trigonal pyramidal trigonal bipyramidal seesaw square pyramidal trigonal planar tetrahedral square planar
Select all molecular geometries (shapes) that are possible for a molecule with five electron groups an the central atom.
Do not include molecular geometries that can only be produced by having a single bonding group on the central atom (all but one of the electron groups are lone pairs.)
| ocahedral | |
| T-shaped | |
| linear | |
| bent | |
| trigonal pyramidal | |
| trigonal bipyramidal | |
| seesaw | |
| square pyramidal | |
| trigonal planar | |
| tetrahedral | |
| square planar |
Reid WolffLv2
13 Dec 2019