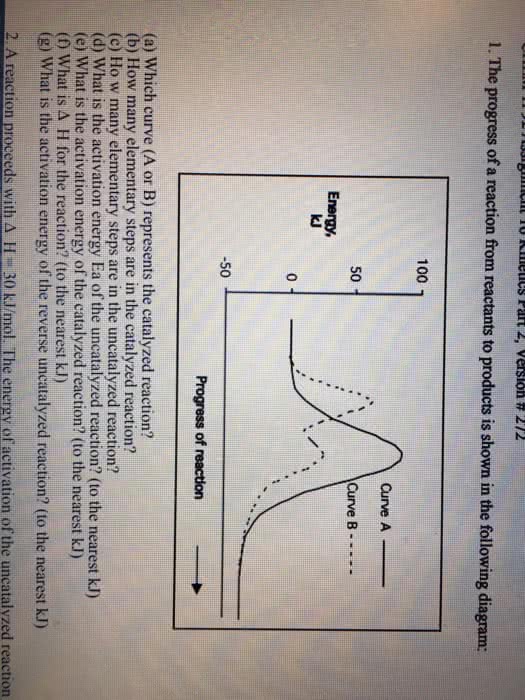
1
answer
0
watching
305
views
roseswan139Lv1
15 Sep 2018
An uncatalyzed reaction, 

A. The activation energy of the reverse reaction is greater than that of the forward reaction.
B. The activation energy of the reverse reaction is lower than that of the forward reaction.
C. If a catalyst is added, the value of 

D. If a catalyst is added, the value of 
E. Since the magnitude of 
An uncatalyzed reaction, 

A. The activation energy of the reverse reaction is greater than that of the forward reaction.
B. The activation energy of the reverse reaction is lower than that of the forward reaction.
C. If a catalyst is added, the value of 

D. If a catalyst is added, the value of 
E. Since the magnitude of 
2 Jun 2021