1
answer
0
watching
413
views
12 Dec 2019
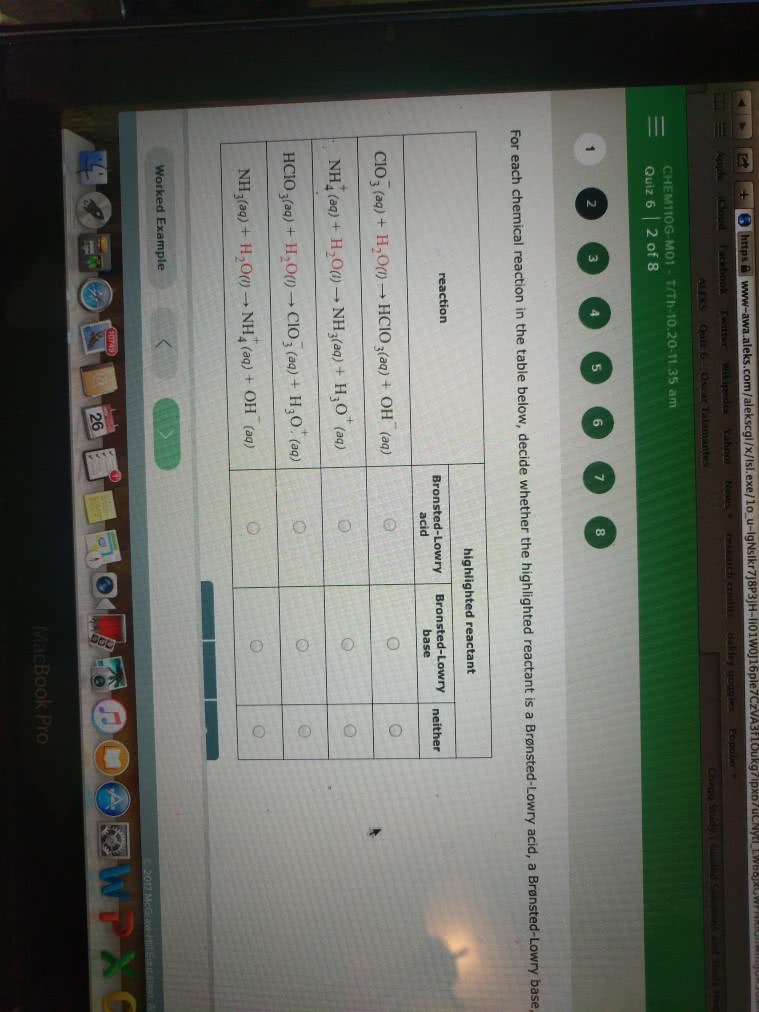
With Acid/Base Theories. For the following 3 reactions identify the reactants that are Athenius bases, Bronsted-Lowry and/ lewis bases:
State which type(s) of bases each reactant is, and be sure to explain your answers.
a. NaOH(s) â Na+(aq) + OH-(aq)
b. HF(aq ) + H2O(l) â F-(aq ) + H3O+(aq )
c. H+(aq ) + NH3(aq ) â NH4+(aq )
With Acid/Base Theories. For the following 3 reactions identify the reactants that are Athenius bases, Bronsted-Lowry and/ lewis bases:
State which type(s) of bases each reactant is, and be sure to explain your answers.
a. NaOH(s) â Na+(aq) + OH-(aq)
b. HF(aq ) + H2O(l) â F-(aq ) + H3O+(aq )
c. H+(aq ) + NH3(aq ) â NH4+(aq )
Jean KeelingLv2
13 Dec 2019