1
answer
0
watching
193
views
12 Dec 2019
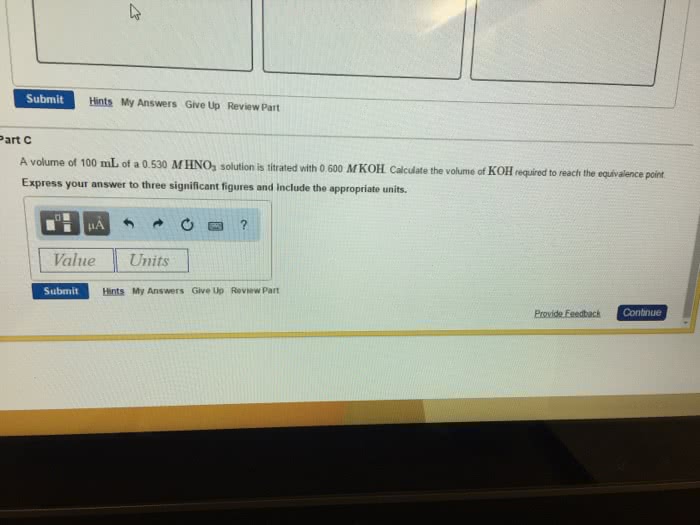
A volume of 90.0 mL of a 0.820 M HNO3 solution is titrated with 0.220 M KOH. Calculate the volume of KOH required to reach the equivalence point.
Express your answer to three significant figures and include the appropriate units.
A volume of 90.0 mL of a 0.820 M HNO3 solution is titrated with 0.220 M KOH. Calculate the volume of KOH required to reach the equivalence point.
Express your answer to three significant figures and include the appropriate units.
Tod ThielLv2
13 Dec 2019