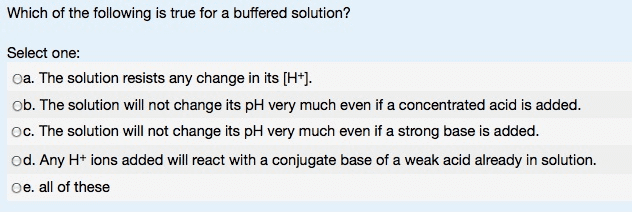
1
answer
0
watching
139
views
12 Dec 2019
1. A 0.100 M solution of the weak acid HB has a pH of 3.00. What are the [H+], [B-], and Ka for this acid?
2. A solid acid is dissolved in water. Half the solution is titrated to a phenolphthalein end point with NaOH solution. The neutralized and acid solutions are then mixed and the pH of the resulting solution is found to be 4.60. Find Ka of the solid acid.
3. Assuming your buffered solution contains acetic acid, HOAc, and the sodium salt of its conjugate base, NaOAc. Show the net ionic equations that occur when this buffered solution reacts with: (a) added strong acid, H+, or (b) added strong base, OH-
1. A 0.100 M solution of the weak acid HB has a pH of 3.00. What are the [H+], [B-], and Ka for this acid?
2. A solid acid is dissolved in water. Half the solution is titrated to a phenolphthalein end point with NaOH solution. The neutralized and acid solutions are then mixed and the pH of the resulting solution is found to be 4.60. Find Ka of the solid acid.
3. Assuming your buffered solution contains acetic acid, HOAc, and the sodium salt of its conjugate base, NaOAc. Show the net ionic equations that occur when this buffered solution reacts with: (a) added strong acid, H+, or (b) added strong base, OH-
Lelia LubowitzLv2
13 Dec 2019