0
answers
0
watching
182
views
12 Dec 2019
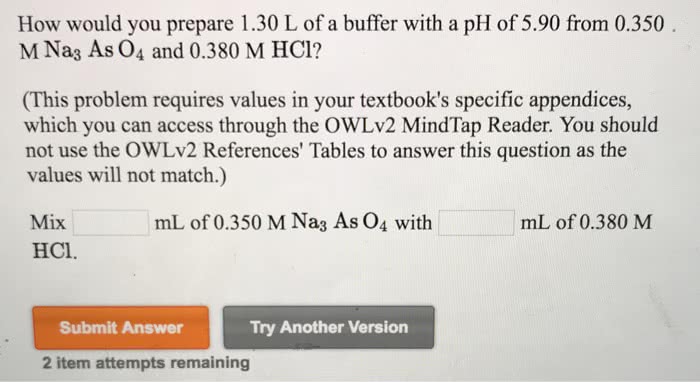
Calculate the pH in the titration of 50.00 mL of 0.1000 M propanoic acid after addition of
(This problem requires values in your textbook's specific appendices, which you can access through the OWLv2 MindTap Reader. You should not use the OWLv2 References' Tables to answer this question as the values will not match. Use a spreadsheet to complete this question.)
0.00 mL of 0.1000 M NaOH
ph=
10.00 mL of 0.1000 M NaOH
ph=
25.00 mL of 0.1000 M NaOH
ph=
40.00 mL of 0.1000 M NaOH
ph=
45.00 mL of 0.1000 M NaOH
ph=
49.00 mL of 0.1000 M NaOH
ph=
50.00 mL of 0.1000 M NaOH
ph=
51.00 mL of 0.1000 M NaOH
ph=
55.00 mL of 0.1000 M NaOH
ph=
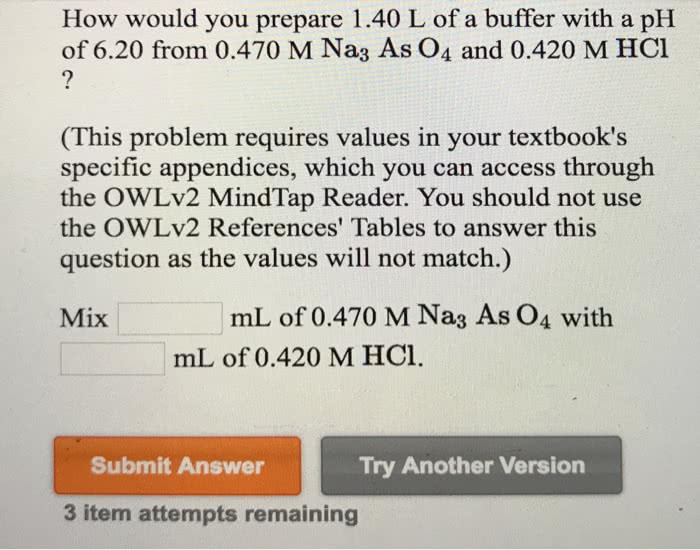
Calculate the pH in the titration of 50.00 mL of 0.1000 M propanoic acid after addition of
(This problem requires values in your textbook's specific appendices, which you can access through the OWLv2 MindTap Reader. You should not use the OWLv2 References' Tables to answer this question as the values will not match. Use a spreadsheet to complete this question.)
0.00 mL of 0.1000 M NaOH
ph=
10.00 mL of 0.1000 M NaOH
ph=
25.00 mL of 0.1000 M NaOH
ph=
40.00 mL of 0.1000 M NaOH
ph=
45.00 mL of 0.1000 M NaOH
ph=
49.00 mL of 0.1000 M NaOH
ph=
50.00 mL of 0.1000 M NaOH
ph=
51.00 mL of 0.1000 M NaOH
ph=
55.00 mL of 0.1000 M NaOH
ph=