1
answer
0
watching
643
views
12 Dec 2019
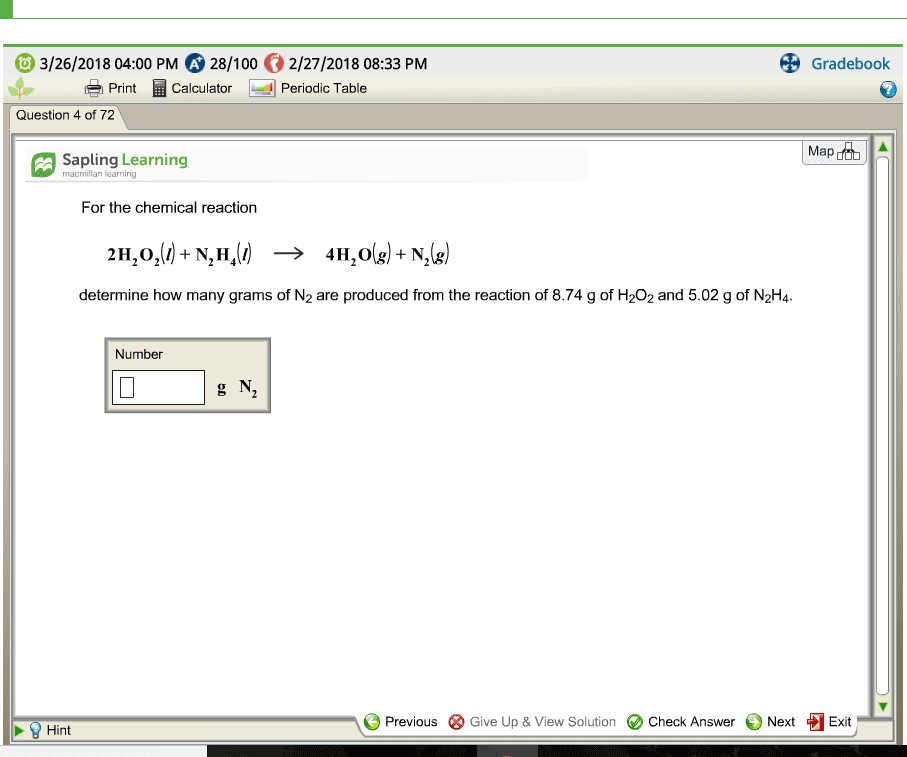
For the chemical reaction
2H2O2(l) +N2H4(l) -> 4H2O(g) + N2(g)
determine how many grams of N2 are produced from the reaction of 8.82 g of H2O2 and 5.63 g of N2H4.
For the chemical reaction
2H2O2(l) +N2H4(l) -> 4H2O(g) + N2(g)
determine how many grams of N2 are produced from the reaction of 8.82 g of H2O2 and 5.63 g of N2H4.
Jean KeelingLv2
13 Dec 2019