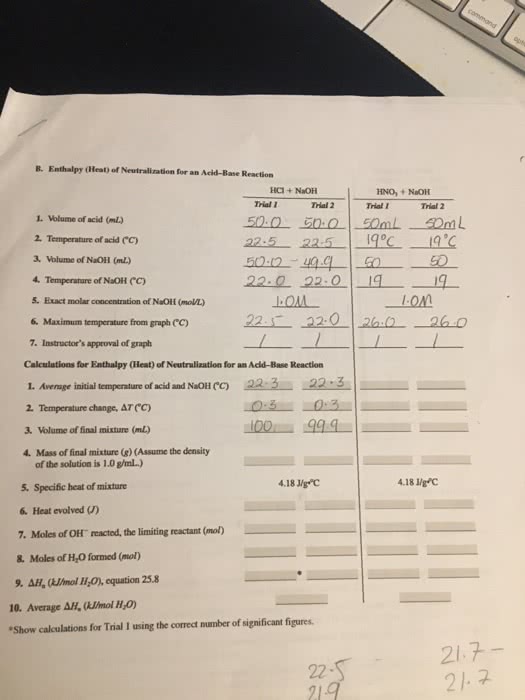
coffee cup calorimeter contained 50.0 mL of a 1.33 M KOH solution at an initial temperature of 23.3 ºC. A student quickly add 40.0 mL of 1.54 M HNO3 to the calorimeter. The maximum temperature reached during the neutralization reaction was 29.1ºC.
assume specific heat of solution is the same as specific heat of water = 4.18 J/gºC
assume density of both KOH and HNO3 are the same as water = 1.00 g/mL
_________________________________________________________________
This is more of a question regarding the question itself.
What's the difference in finding the mass using Mass = moles*Molar mass = (MolarityÃVolume)*Molar mass
and finding mass using Mass = Density*Volume
There is obviously a big difference in these values.
For example, finding the mass of KOH using molarity and volume
Mass = (1.33)*(50*10^-3)*56.105 = 3.73g
Using density it'll be Mass = 50* 1 = 50g
What's the difference, and which method is the most correct?
coffee cup calorimeter contained 50.0 mL of a 1.33 M KOH solution at an initial temperature of 23.3 ºC. A student quickly add 40.0 mL of 1.54 M HNO3 to the calorimeter. The maximum temperature reached during the neutralization reaction was 29.1ºC.
assume specific heat of solution is the same as specific heat of water = 4.18 J/gºC
assume density of both KOH and HNO3 are the same as water = 1.00 g/mL
_________________________________________________________________
This is more of a question regarding the question itself.
What's the difference in finding the mass using Mass = moles*Molar mass = (MolarityÃVolume)*Molar mass
and finding mass using Mass = Density*Volume
There is obviously a big difference in these values.
For example, finding the mass of KOH using molarity and volume
Mass = (1.33)*(50*10^-3)*56.105 = 3.73g
Using density it'll be Mass = 50* 1 = 50g
What's the difference, and which method is the most correct?