1
answer
0
watching
367
views
11 Dec 2019
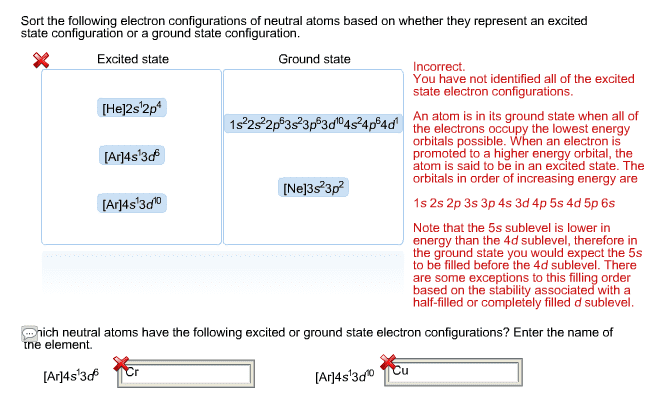
What is the ground state electron configuration and orbital diagram of Os(76)? Is this paramagnetic or diamagnetic?
(b) The following is an excited state configuration of a neutral atom: 1s22s22p53s1 3p44s2 5s26s15p5 Identify the element and write its ground state electronic configuration. Is this excited state diamagnetic or paramagnetic? Explain
(c) 8pts, Write the ground state electron configuration of each of the following ions: (i)Mn4+(25) (ii) In5+(49) (iii) Hg2+(80) (iv) Sb3-(51)
What is the ground state electron configuration and orbital diagram of Os(76)? Is this paramagnetic or diamagnetic?
(b) The following is an excited state configuration of a neutral atom: 1s22s22p53s1 3p44s2 5s26s15p5 Identify the element and write its ground state electronic configuration. Is this excited state diamagnetic or paramagnetic? Explain
(c) 8pts, Write the ground state electron configuration of each of the following ions: (i)Mn4+(25) (ii) In5+(49) (iii) Hg2+(80) (iv) Sb3-(51)
Deanna HettingerLv2
13 Dec 2019