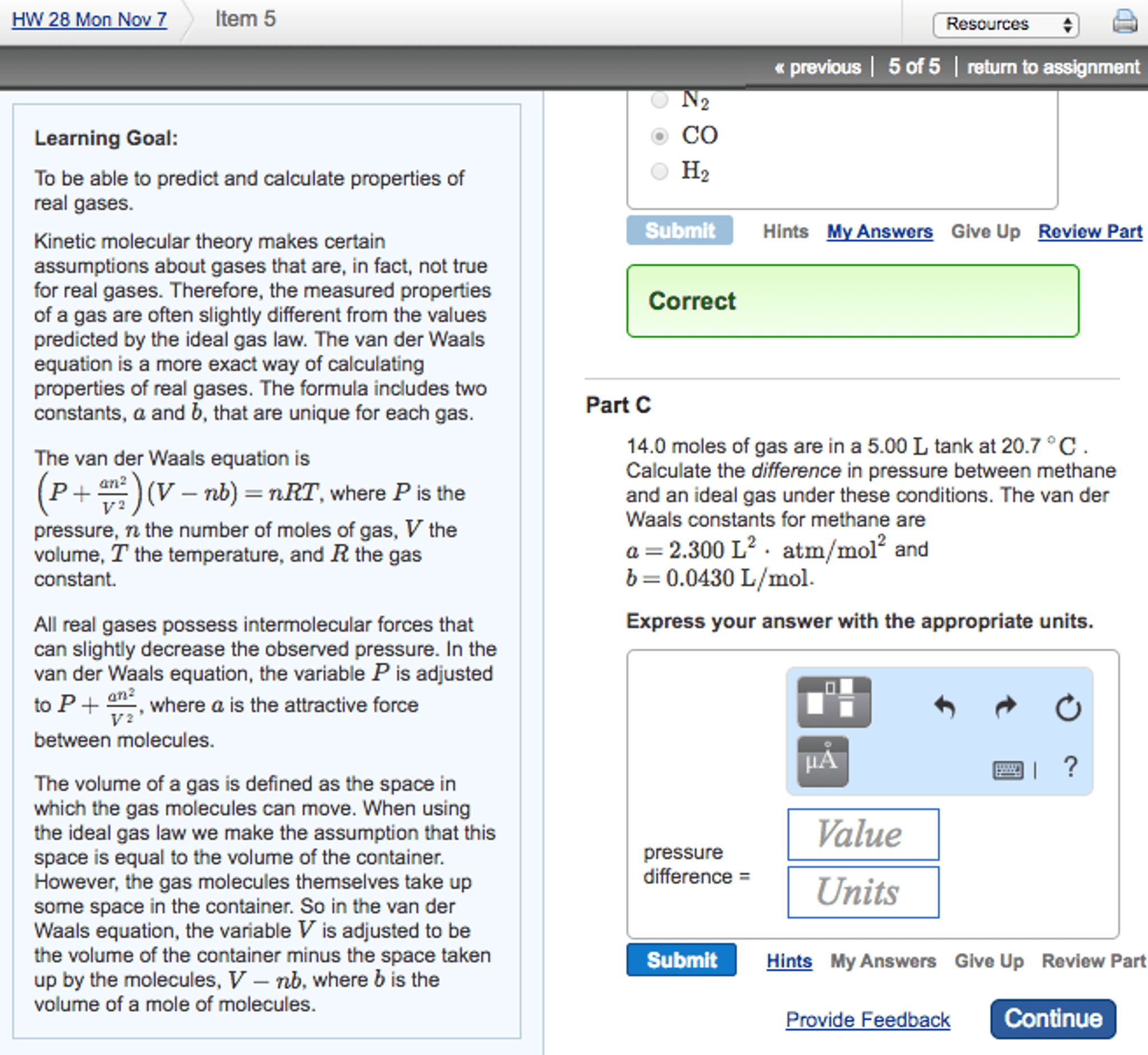
1
answer
0
watching
97
views
11 Dec 2019
Which of the following statements is false ?
A for one mole of any gas behaving ideally, the ratio PV/RT = 1.0 B the van der Waals equation adjusts the volume down to account for interparticular repulsion between gas particles at very high pressures and/or very low temperatures
C increasing the pressure of an ideal gas decreases its average kinetic energy and collision frequency D the van der Waals equation adjusts the pressure up to account for interparticular attraction between gas particles at high pressures and/or low temperatures E the average kinetic energy of any sample of gas is the same at the same temperature
Which of the following statements is false ?
| A | for one mole of any gas behaving ideally, the ratio PV/RT = 1.0 | ||
| B |
| ||
| C | increasing the pressure of an ideal gas decreases its average kinetic energy and collision frequency | ||
| D | the van der Waals equation adjusts the pressure up to account for interparticular attraction between gas particles at high pressures and/or low temperatures | ||
| E |
|
Trinidad TremblayLv2
13 Dec 2019