Write the equation for calculating the initial rates of each of these reactions. Rate = Î[I2]/Ît The indicator changed color when all the thiosulfate ions, S2O32-, were used up. Upon the instant the solutions were mixed, you calculated a molarity of S2O32- (from Q 9). Since the concentration of thiosulfate changed from this value to zero this is the value of Î[S2O32-].
What is the value of Î[I2] at the point of the color change for each of the runs? (Show your work) Consider the balanced reaction for the clock step:
I2(aq) + 2 S2O32-(aq) â 2 I-(aq) + S4O62-(aq)
Write the equation for calculating the initial rates of each of these reactions. Rate = Î[I2]/Ît The indicator changed color when all the thiosulfate ions, S2O32-, were used up. Upon the instant the solutions were mixed, you calculated a molarity of S2O32- (from Q 9). Since the concentration of thiosulfate changed from this value to zero this is the value of Î[S2O32-].
What is the value of Î[I2] at the point of the color change for each of the runs? (Show your work) Consider the balanced reaction for the clock step:
I2(aq) + 2 S2O32-(aq) â 2 I-(aq) + S4O62-(aq)
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
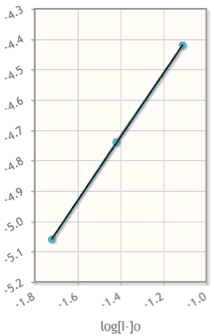
Everytime I make the graph based off my calculations in the chart it looks like this and I have no idea if it is even close? Please help ! For the questions I just need to understand how to start them and what info is nessiary to do so. very confused. 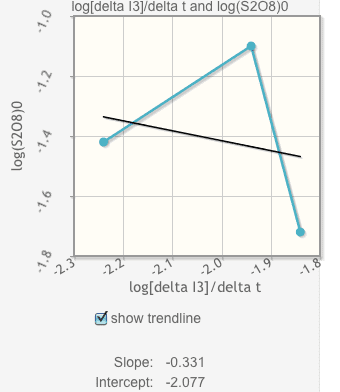
Data Analysis
Fill in the following table. Look below the table for instructions on how to calculate the values for each row of the table.
| a | total volume (L) | 0.065L |
| b | mol S2O32-consumed | 0.010 mol |
| c | mol I3-produced | 0.015M |
| d | [S2O32-] consumed (M) | 0.154M |
| e | [I3-] produced (M) | 0.231M |
Calculate the final volume of the reaction mixture after the contents of beaker B are added to beaker A. Report your answer in liters.
All S2O32- is consumed at the end of the reaction. Therefore, the moles of S2O32- consumed can be calculated using the equation below.
(moles S2O32-)consumed = Mstock x (Vstock)
The I3- produced in reaction 1 reacts with S2O32- in reaction 2 as shown.
I3- (aq) 2S2O32- (aq) â 3I- (aq) S4O62- (aq)
Therefore, for every mole of I3- produced, 2 moles of S2O32- have reacted.
(moles I3-)produced = (moles S2O32-)consumed / 2
[S2O32-]consumed = (moles S2O32-)consumed / (Vtotal)
Calculation is the same as d, but using I3- instead of S2O32-.
Fill in the following table. Look below the table for instructions on how to calculate the values for each column of the table.
| a | b | c | d | e | f | g | h | i | |
|---|---|---|---|---|---|---|---|---|---|
| Trial | Ît | Î[I3-] / Ît (M/s) | log(Î[I3-] / Ît) | V 0.2 M KI added (mL) | [Iâ]0(M) | log[Iâ]0 | V of 0.2 M (NH4)S2O8 (mL) | [S2O8]0 (M) | log[S2O8]0 |
| 1 | 19 | .011 | -1.94 | 25 | .077 | -1.35 | 25 | .077 | -1.11 |
| 2 | 40 | .0055 | -2.24 | 25 | .077 | -1.11 | 12.5 | .038 | -1.42 |
| 3 | 85 | .0030 | -1.84 | 25 | .077 | -.1.11 | 6.25 | .019 | -1.72 |
| 4 | 42 | .0055 | -2.26 | 12.5 | .038 | -1.42 | 25 | .077 | -1.11 |
Conclusions
If a large amount of heat was released at the start of the reaction, what effect would this have on the rate measurements?
| The rate of reaction would be much slower due to its exothermic reaction properties. |
Click the box underneath the graph to show the trendline. It will automatically calculate your slope and intercept. Record them below.
| Determine the value of q from the graph. Explain your answer. |
What is the rate law for the reaction?
For each trial, calculate the rate constant. What is average value of the rate constant?
In this experiment, you assumed that [S2O82â] >> [S2O32â]. To find out if this assumption is correct, calculate the ratio of [S2O82â] / [S2O32â] for Trial 3. (In Trial 3, the [S2O82â] was lowest, and therefore the ratio [S2O82â] / [S2O32â] is the smallest).
In this experiment, you assumed that only a small amount of S2O82â was used during the time trial so that the concentration of this reactant did not change appreciably during the course of the reaction. Calculate how much S2O82â was used in Trial 3 and the percent remaining at the end of the reaction. Was this assumption valid?
The kinetics of the reaction below were studied. The volumes of 0.040 M KBrO3, 0.040 M KI,
0.00050 M Na2S2O3, 0.040 M KCl, H2O and a buffer solution employed in each trial are shown
in the table below. One drop of 1% aqueous starch solution was also added in each trial. The
buffer solution was a pH = 4.74 solution containing 0.50 M acetic acid and 0.50 M sodium acetate; Ka = 1.8 x 10â5 for acetic acid. Each trial was performed at 25â¦C. For each trial, the reaction mixture was initially colorless after combining all solutions; appearance of a blue solution color in the reaction mixture signaled that all of the thiosulfate ion initially present had reacted and marked the end of the timing period. The initial reaction rate for each trial was calculated from the change in molar concentration of thiosulfate ion from each trial; the equation for calculating Initial Rate is shown below, where Ît is the elapsed time between combining all solutions and appearance of the blue solution color. Each solution volume was measured to the nearest 0.1 mL. (ie 5 mL shown in the table is 5.0 mL.)
BrO3- (aq) + 6Iâ(aq) + 6H+(aq) â 3H2O(l) + Brâ(aq) + 3I2(aq)
Initial rate = - 13âS2O32-ât
Volume (mL) Used
| Trial # | KBrO3 | KI | Na2S2O3 | KCl | H2O | Buffer | Initial Rate (M/s) |
| 1 | 10 | 10 | 5 | 0 | 0 | 15 | 5.0 x 10â6 |
| 2 | 5 | 10 | 5 | 5 | 0 | 15 | 2.5 x 10â6 |
| 3 | 10 | 5 | 5 | 5 | 0 | 15 | 2.5 x 10â6 |
| 4 | 10 | 10 | 2.5 | 0 | 2.5 | 15 | 5.0 x 10â6 |
a)Which pair of trials could be used to determine the order for Iâ? Justify your choice.
b)How would you determine the order for H+? Be as specific as possible.
c)What would be the value for the initial rate if 5 mL KBrO3 solution, 5 mL KI solution, 5 mL Na2S2O3 solution, 10 mL KCl solution, 15 mL pH = 4.74 buffer solution, and 1 drop of 1% starch solution were used?
d)Why do Trials 1 and 4 have the same initial rate?
e)What time was required for the color change to occur in Trial 1?
f)What was the purpose of adding 5 mL of 0.040 M KCl in Trial 2?
g)How would you experimentally verify that KCl does not cause the color change from colorless to blue? Be as specific as possible.