1
answer
0
watching
144
views
11 Dec 2019
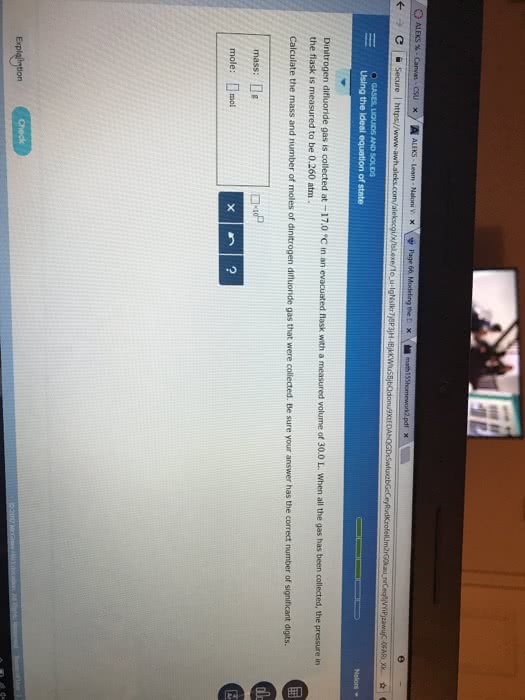
Carbon dioxide gas is collected at -5 degrees C in an evacuated flask with a measured volume of 20 Liters. When all the gas has been collected, the pressure in the flask is measured to be .420 atm. Calculate the mass(g) and number of moles of carbon dioxide gas that were collected. Round your answer to 3 sig figs. answers are not .368 moles and 16.1 g
Carbon dioxide gas is collected at -5 degrees C in an evacuated flask with a measured volume of 20 Liters. When all the gas has been collected, the pressure in the flask is measured to be .420 atm. Calculate the mass(g) and number of moles of carbon dioxide gas that were collected. Round your answer to 3 sig figs. answers are not .368 moles and 16.1 g
Nelly StrackeLv2
13 Dec 2019