1
answer
0
watching
151
views
11 Dec 2019
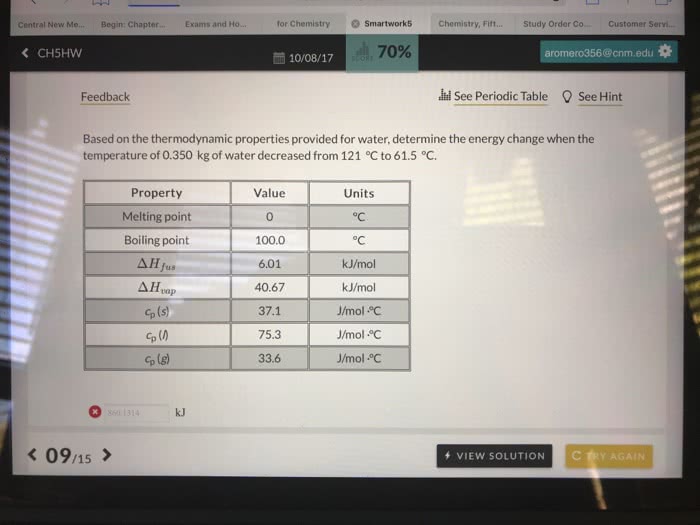
The ÎH°soln of HNO3 is â33.3 kJ/mol. 10.0 mL of 12.0 M HNO3 is dissolved in 100.0 mL of distilled water initially at 25°C.
How much ice at 0°C [Cp = 37.1 J/(mol ·°C), ÎH°fus = 6.01 kJ/mol] must be added to return the solution temperature to 25°C after dissolution of the acid and equilibrium with the ice is reached? The molar heat capacity is 80.8 J/(mol·°C) for the solution, and the molar heat capacity is 75.3 J/(mol·°C) for pure water.
The ÎH°soln of HNO3 is â33.3 kJ/mol. 10.0 mL of 12.0 M HNO3 is dissolved in 100.0 mL of distilled water initially at 25°C.
How much ice at 0°C [Cp = 37.1 J/(mol ·°C), ÎH°fus = 6.01 kJ/mol] must be added to return the solution temperature to 25°C after dissolution of the acid and equilibrium with the ice is reached? The molar heat capacity is 80.8 J/(mol·°C) for the solution, and the molar heat capacity is 75.3 J/(mol·°C) for pure water.
Trinidad TremblayLv2
13 Dec 2019