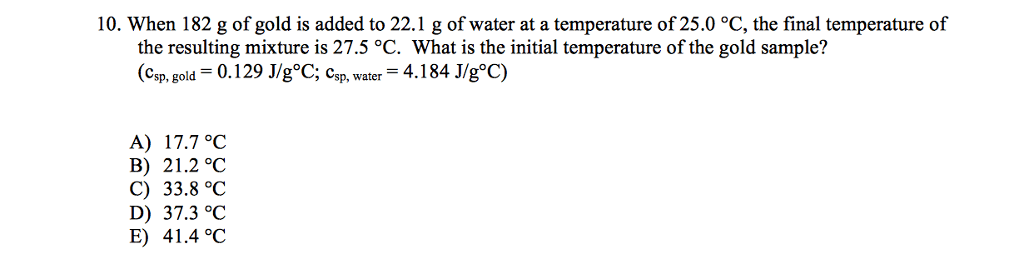1
answer
0
watching
111
views
11 Dec 2019
When 182 g of gold at some temperature is placed in 25 mL of water at 25.0 oC the final temperature of the mixture is 28.3 oC. What was the initial temperature of the gold? (CAu = 0.128 J/gK)
When 182 g of gold at some temperature is placed in 25 mL of water at 25.0 oC the final temperature of the mixture is 28.3 oC. What was the initial temperature of the gold? (CAu = 0.128 J/gK)
Patrina SchowalterLv2
13 Dec 2019