0
answers
0
watching
174
views
11 Dec 2019
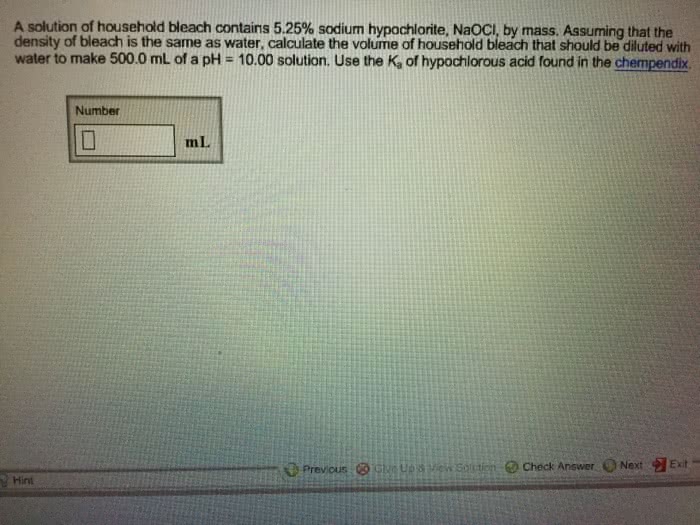
1) A solution of household bleach contains 5.25% sodium hypochlorite, NaOCl, by mass. Assuming that the density of bleach is the same as water, calculate the volume of household bleach that should be diluted with water to make 500.0 mL of a pH = 9.80 solution. Use the Ka of hypochlorous acid found in the chempendix
2) For the diprotic weak acid H2A, Ka1 = 2.5 Ã 10-6 and Ka2 = 8.0 Ã 10-9. What is the pH of a 0.0800 M solution of H2A? What are the equilibrium concentrations of H2A and A2â in this solution?
1) A solution of household bleach contains 5.25% sodium hypochlorite, NaOCl, by mass. Assuming that the density of bleach is the same as water, calculate the volume of household bleach that should be diluted with water to make 500.0 mL of a pH = 9.80 solution. Use the Ka of hypochlorous acid found in the chempendix
2) For the diprotic weak acid H2A, Ka1 = 2.5 Ã 10-6 and Ka2 = 8.0 Ã 10-9. What is the pH of a 0.0800 M solution of H2A? What are the equilibrium concentrations of H2A and A2â in this solution?