1
answer
0
watching
1,946
views
11 Dec 2019
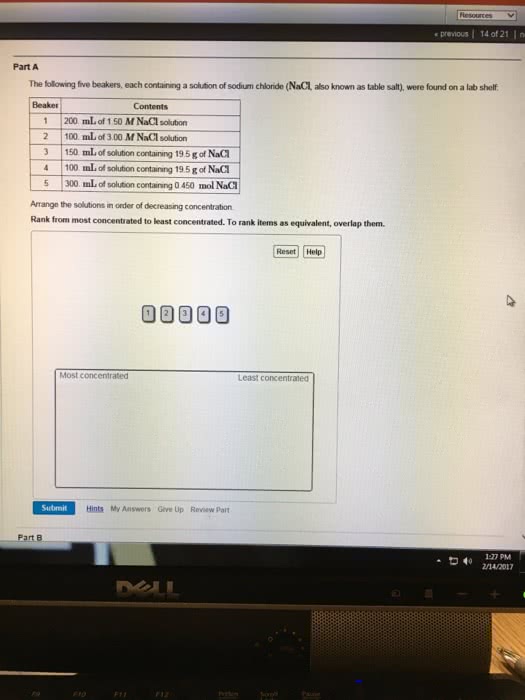
the following five beakers, each containing a solution of sodium chloride (NaCl, also known as table salt), were found on a lab shelf:
Beaker Contents 1 200. mL of 1.50 M NaCl solution 2 100. mL of 3.00 M NaCl solution 3 150. mL of solution containing 25.0 g of NaCl 4 100. mL of solution containing 25.0 g of NaCl 5 300. mL of solution containing 0.450 mol NaCl
Arrange the solutions in order of decreasing concentration.
Rank from most concentrated to least concentrated. To rank items as equivalent, overlap them
the following five beakers, each containing a solution of sodium chloride (NaCl, also known as table salt), were found on a lab shelf:
| Beaker | Contents |
| 1 | 200. mL of 1.50 M NaCl solution |
| 2 | 100. mL of 3.00 M NaCl solution |
| 3 | 150. mL of solution containing 25.0 g of NaCl |
| 4 | 100. mL of solution containing 25.0 g of NaCl |
| 5 | 300. mL of solution containing 0.450 mol NaCl |
Arrange the solutions in order of decreasing concentration.
Rank from most concentrated to least concentrated. To rank items as equivalent, overlap them
Elin HesselLv2
13 Dec 2019