1
answer
0
watching
1,695
views
11 Dec 2019
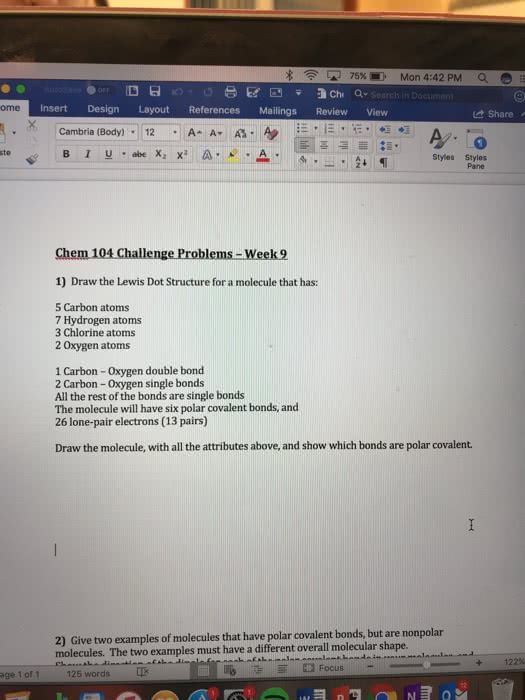
Please answer all questions
1. Which type of chemical bond involves the transfer of electrons?
A. Ionic
B. Polar covalent
C. Nonpolar covalent
2. A polar covalent bond will form between which two atoms?
A. Beryllium and fluorine (group 1 and group 7)
B. Hydrogen and chlorine (group 7)
C. Sodium and oxygen (group 1 and group 6)
D. Fluorine and fluorine (group 7)
3. A nonpolar covalent bond will form between which two atoms?
A. Potassium and chlorine
B. Hydrogen and chlorine
C. Sodium and bromine
D. Chlorine and chlorine
4. As the difference in electronegativity between two atoms increases, the polarity of a bond between those two atoms
A. increases.
B. decreases.
C. remains the same.
Please answer all questions
1. Which type of chemical bond involves the transfer of electrons?
A. Ionic
B. Polar covalent
C. Nonpolar covalent
2. A polar covalent bond will form between which two atoms?
A. Beryllium and fluorine (group 1 and group 7)
B. Hydrogen and chlorine (group 7)
C. Sodium and oxygen (group 1 and group 6)
D. Fluorine and fluorine (group 7)
3. A nonpolar covalent bond will form between which two atoms?
A. Potassium and chlorine
B. Hydrogen and chlorine
C. Sodium and bromine
D. Chlorine and chlorine
4. As the difference in electronegativity between two atoms increases, the polarity of a bond between those two atoms
A. increases.
B. decreases.
C. remains the same.
Casey DurganLv2
13 Dec 2019