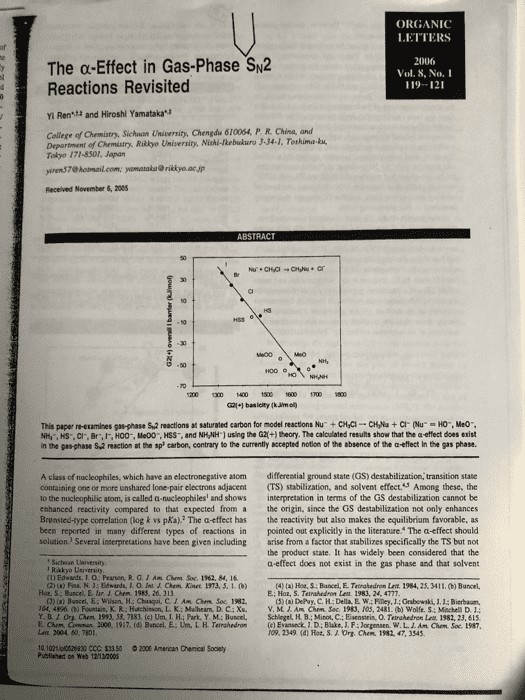
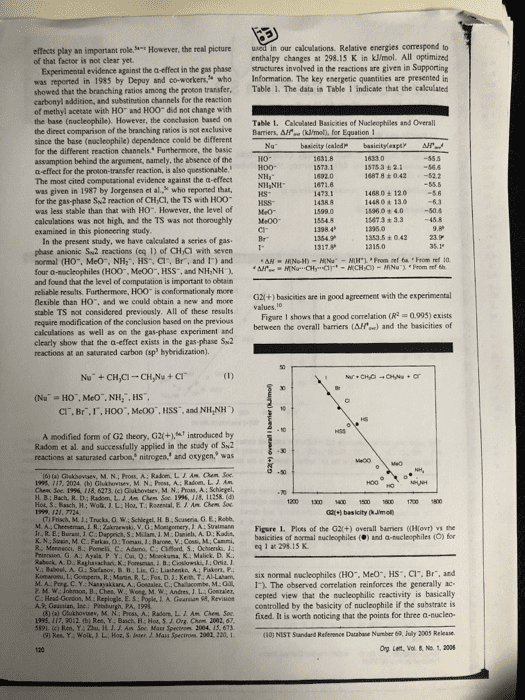
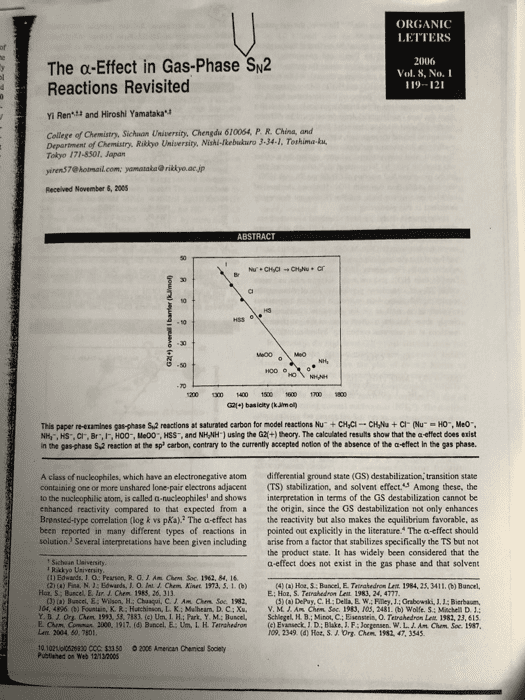
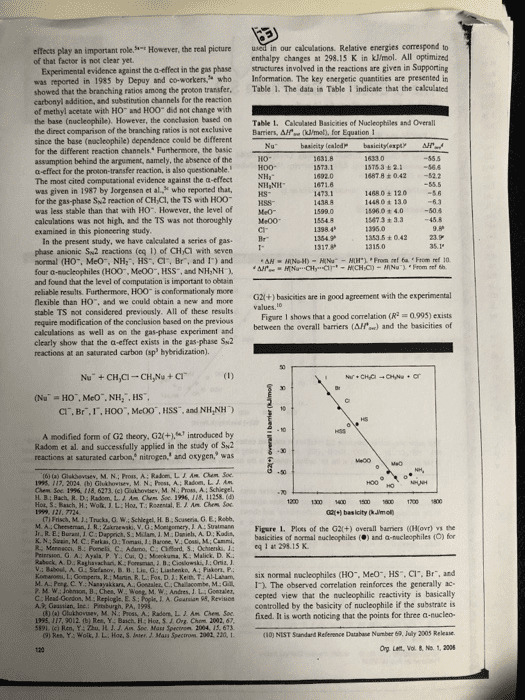
1
answer
0
watching
102
views
11 Dec 2019
In an organic chemistry lab we were looking at the substrate and nucleophile effects in Sn2 reactions. Our experimental data showed that 1-bromobutane with 1.0mL of 0.25M NaI in 2-Butanone reacted faster than 1-bromobutane with 1.0mL 0.50M NaI in 2-butanone. Also, our experimental data showed that 1-bromobutane with 1.0mL of 0.50M NaI reacted faster than 1-bromobutane with 2.0mL of 0.05M NaI.
These result are the opposite of the theoritcal rate aren't they? From my understanding, the Sn2 reaction rate should should double when the nucleophile or substarte is doubled.
In an organic chemistry lab we were looking at the substrate and nucleophile effects in Sn2 reactions. Our experimental data showed that 1-bromobutane with 1.0mL of 0.25M NaI in 2-Butanone reacted faster than 1-bromobutane with 1.0mL 0.50M NaI in 2-butanone. Also, our experimental data showed that 1-bromobutane with 1.0mL of 0.50M NaI reacted faster than 1-bromobutane with 2.0mL of 0.05M NaI.
These result are the opposite of the theoritcal rate aren't they? From my understanding, the Sn2 reaction rate should should double when the nucleophile or substarte is doubled.
1
answer
0
watching
102
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Keith LeannonLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232