1
answer
0
watching
122
views
11 Dec 2019
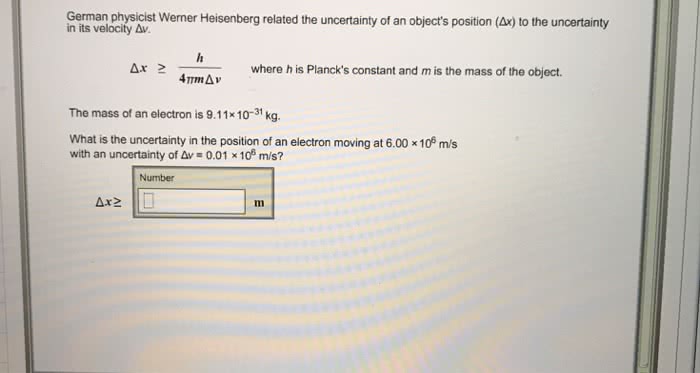
Heisenberg's uncertainty principle can be expressed mathematically as Îx*Îp = h/4Ï, where Îx and Îp denote the uncertainty in position and momentum respectively and h is Planck's constant. What would be the uncertaintry in the position of a snail (mass = 0.005178 kg) that was traveling at a velocity of 0.00004313 m/s if the velocity has an uncertainty of 2.368%?
Heisenberg's uncertainty principle can be expressed mathematically as Îx*Îp = h/4Ï, where Îx and Îp denote the uncertainty in position and momentum respectively and h is Planck's constant. What would be the uncertaintry in the position of a snail (mass = 0.005178 kg) that was traveling at a velocity of 0.00004313 m/s if the velocity has an uncertainty of 2.368%?
Lelia LubowitzLv2
13 Dec 2019