1
answer
0
watching
86
views
11 Dec 2019
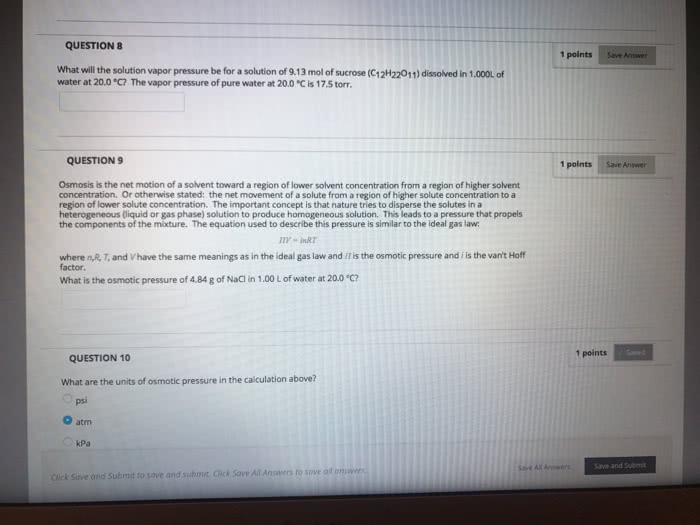
Osmotic pressure is the
a. pressure that must be applied to a solution in order to prevent osmosis from the pure solvent.
b. pressure that must be applied to a solution in order to cause osmosis from the pure solvent.
c. correction factor applied when a sample of gas is collected by water displacement.
d. increase in vapor pressure when a solution is compared to a pure solvent.
e. decrease in vapor pressure when a solution is compared to a pure solvent.
Osmotic pressure is the
a. pressure that must be applied to a solution in order to prevent osmosis from the pure solvent.
b. pressure that must be applied to a solution in order to cause osmosis from the pure solvent.
c. correction factor applied when a sample of gas is collected by water displacement.
d. increase in vapor pressure when a solution is compared to a pure solvent.
e. decrease in vapor pressure when a solution is compared to a pure solvent.
Hubert KochLv2
13 Dec 2019